Calcium acetate
Synonym(s):Calcium acetate
- CAS NO.:62-54-4
- Empirical Formula: C4H6CaO4
- Molecular Weight: 158.17
- MDL number: MFCD00012448
- EINECS: 200-540-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-05-29 16:45:47
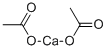
What is Calcium acetate?
Absorption
40% is absorbed in the fasting state and approximately 30% is absorbed in the nonfasting state following oral administration.
Toxicity
Oral, rat: LD50 = 4280 mg/kg. Symptoms of overdose include mild hypercalcemia (constipation; loss of appetite; nausea and vomiting), and severe hypercalcemia (confusion; full or partial loss of consciousness; incoherent speech).
Description
Calcium acetate is a chemical compound which is calcium salt of acetic acid. It has the formula Ca(C2H3O2)2. Its standard name is calcium acetate, while calcium ethanoate is the systematic name. An older name is acetate of lime. The anhydrous form is very hygroscopic; therefore the monohydrate (Ca(CH3COO)2?H2O) is the common form.
The Uses of Calcium acetate
Calcium Acetate is the calcium salt of acetic acid which functions as a sequestrant and mold control agent. it contains approximately 25% calcium. it is a white odorless powder which is readily soluble in water with a solubility of approximately 37 g in 100 g water at 0°c. its solubility decreases with increasing temperature, with a sol- ubility of approximately 29 g in 100 g of water at 100°c.
Indications
Calcium acetate is one of a number of calcium salts used to treat hyperphosphatemia (too much phosphate in the blood) in patients with kidney disease.
Background
The chemical compound calcium acetate is the calcium salt of acetic acid. It has been commonly referred to as the acetate of lime. The anhydrous form is very hygroscopic, therefore the monohydrate is the common form.
Pharmacokinetics
Patients with advanced renal insufficiency (creatinine clearance less than 30 ml/min) exhibit phosphate retention and some degree of hyperphosphatemia. The retention of phosphate plays a pivotal role in causing secondary hyperparathyroidism associated with osteodystrophy, and soft-tissue calcification. The mechanism by which phosphate retention leads to hyperparathyroidism is not clearly delineated. Therapeutic efforts directed toward the control of hyperphosphatemia include reduction in the dietary intake of phosphate, inhibition of absorption of phosphate in the intestine with phosphate binders, and removal of phosphate from the body by more efficient methods of dialysis. The rate of removal of phosphate by dietary manipulation or by dialysis is insufficient. Dialysis patients absorb 40% to 80% of dietary phosphorus. Therefore, the fraction of dietary phosphate absorbed from the diet needs to be reduced by using phosphate binders in most renal failure patients on maintenance dialysis. Calcium acetate when taken with meals combines with dietary phosphate to form insoluble calcium phosphate which is excreted in the feces. Maintenance of serum phosphorus below 6.0 mg/dl is generally considered as a clinically acceptable outcome of treatment with phosphate binders. Calcium acetate is highly soluble at neutral pH, making the calcium readily available for binding to phosphate in the proximal small intestine.
Metabolism
Not Available
Properties of Calcium acetate
| Melting point: | 160°C (dec.) |
| Density | 1,5 g/cm3 |
| Flash point: | 160°C |
| storage temp. | Hygroscopic, Room Temperature, under inert atmosphere |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| form | Powder |
| color | white |
| Water Solubility | soluble |
Safety information for Calcium acetate
Computed Descriptors for Calcium acetate
| InChIKey | VSGNNIFQASZAOI-UHFFFAOYSA-L |
Abamectin manufacturer
SAGAR LIFE SCIENCES PVT LTD
Mahadev Pharmaceuticals
Evans Fine Chem
UNILOSA INTERNATINAL PRIVATE LIMITED
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran

You may like
-
 CALCIUM ACETATE USP 99%View Details
CALCIUM ACETATE USP 99%View Details -
 CALCIUM ACETATE 99%View Details
CALCIUM ACETATE 99%View Details -
 CALCIUM DI ACETATE 99%View Details
CALCIUM DI ACETATE 99%View Details -
 Calcium Acetate 99%View Details
Calcium Acetate 99%View Details -
 Calcium acetate 99%View Details
Calcium acetate 99%View Details -
 Calcium acetate 99%View Details
Calcium acetate 99%View Details -
 Calcium acetate 98%View Details
Calcium acetate 98%View Details -
 Calcium acetate 99%View Details
Calcium acetate 99%View Details