Cabazitaxel
Synonym(s):;DEP cabazitaxel;dimethoxydocetaxel;Jevtana;
- CAS NO.:183133-96-2
- Empirical Formula: C45H57NO14
- Molecular Weight: 835.93
- MDL number: MFCD18827611
- EINECS: 680-632-7
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-05-01 11:53:09
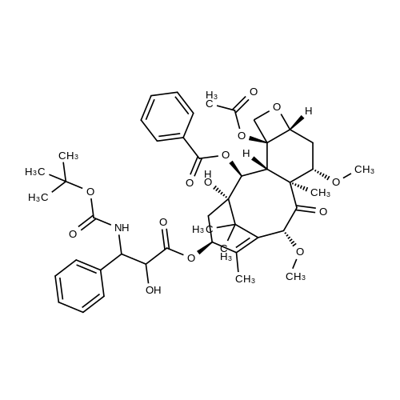
What is Cabazitaxel?
Absorption
Based on the population pharmacokinetic analysis, after an intravenous dose of cabazitaxel 25 mg/m2 every three weeks, the mean Cmax in patients with metastatic prostate cancer was 226 ng/mL (CV 107%) and was reached at the end of the one-hour infusion (Tmax). The mean AUC in patients with metastatic prostate cancer was 991 ng x h/mL (CV 34%). No major deviation from the dose proportionality was observed from 10 to 30 mg/m2 in patients with advanced solid tumours.
Toxicity
The oral LD50 in rats is 500 mg/kg.
Description
In June 2010, the U.S. FDA approved cabazitaxel (also referred to as
XRP6258 and RPR 116258A) in combination with the steroid prednisone
for the treatment of metastatic Castration-Resistant Prostate Cancer
(mCRPC) for patients who were previously treated with a docetaxelcontaining
regimen for late-stage disease.
Cabazitaxel is a semisynthetic analog of the
natural product taxol, which is isolated from the bark of the yew tree.
Cabazitaxel is a microtubule inhibitor that binds to the taxol-binding site of
tubulin. Similar to other tubulin inhibitors of the taxol class, cabazitaxel
inhibits microtubule disassembly resulting in mitotic blockade and cell
death. Docetaxel, also a semisynthetic taxol analog, was approved by the
FDA for the treatment of mCRPC in 2004. However, docetaxel is a substrate
for P-gp, which is thought to contribute to the constitutive and acquired
resistance of cancer cells to taxanes. Cabazitaxel has poor affinity for P-gp
and showed antitumor activity in preclinical in vitro studies and in vivo
tumor models that overexpress this protein. Cabazitaxel is synthesized on
a commercial scale from 10-deacetylbaccatin .
The Uses of Cabazitaxel
Cabazitaxel (Jevtana, XRP6258) is a semi-synthetic derivative of a natural taxoid.
Indications
Cabazitaxel is indicated, in combination with prednisone, for the treatment of patients with metastatic castration-resistant prostate cancer previously treated with a docetaxel-containing treatment regimen. In Europe and Canada, it can also be used in combination with prednisolone.
Background
Cabazitaxel is a taxoid synthesized from 10-deacetylbaccatin III, a compound isolated from the yew tree. As a second-generation semisynthetic microtubule inhibitor, cabazitaxel stabilizes microtubules and induces tumour cell death. Due to its low affinity for the P-glycoprotein (P-gp) efflux pump, cabazitaxel can more readily penetrate the blood–brain barrier compared to other taxanes like paclitaxel and docetaxel.
Cabazitaxel is used to treat metastatic castration-resistant prostate cancer. It was first approved by the FDA on June 17, 2010. It was also approved by the EMA on March 17, 2011 and Health Canada on December 17, 2019.
What are the applications of Application
Cabazitaxel is a semi-synthetic toxal derivative
Pharmacokinetics
Cabazitaxel demonstrates a broad spectrum of antitumour activity against advanced human tumours xenografted in mice, including intracranial human glioblastomas. Cabazitaxel has a low affinity to P-glycoprotein, allowing it to penetrate the blood-brain barrier without being subject to extensive P-gp-mediated active efflux. Cabazitaxel works against docetaxel-sensitive tumours and tumour models resistant to docetaxel and other chemotherapy drugs.
Metabolism
More than 95% of cabazitaxel is extensively metabolized in the liver. CYP3A4 and CYP3A5 are responsible for 80% to 90% of drug metabolism, while CYP2C8 is involved to a lesser extent. While cabazitaxel is the main circulating moiety in human plasma, seven metabolites have been detected in plasma, including three active metabolites arising from O-demethylation - docetaxel, RPR112698, and RPR123142. The main metabolite accounts for 5% of total cabazitaxel exposure.
Properties of Cabazitaxel
| Melting point: | 180 °C |
| Boiling point: | 870.7±65.0 °C(Predicted) |
| Density | 1.31 |
| storage temp. | Inert atmosphere,Store in freezer, under -20°C |
| solubility | Chloroform (Slightly), DMSO (Slightly), Methanol (Slightly) |
| form | White solid. |
| color | White to Off-White |
Safety information for Cabazitaxel
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07  Health Hazard GHS08 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H341:Germ cell mutagenicity H373:Specific target organ toxicity, repeated exposure |
| Precautionary Statement Codes |
P202:Do not handle until all safety precautions have been read and understood. P260:Do not breathe dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P302+P352:IF ON SKIN: wash with plenty of soap and water. P308+P313:IF exposed or concerned: Get medical advice/attention. |
Computed Descriptors for Cabazitaxel
Abamectin manufacturer
Dr. Reddy's Laboratories Ltd
Shilpa Medicare Limited (SML)
Basil Drugs AND Pharmaceuticals Pvt Ltd
BDR Pharmaceuticals International Pvt Ltd
Shreyaa Medilife Private Limited
Vijaya Pharma And Life Science
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6 Di acetylpyridine 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol 3-NITRO-5-ACETYL IMINODIBENZYL (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran

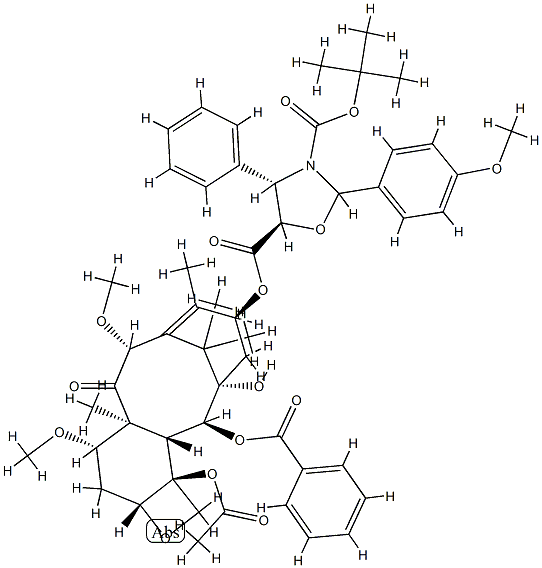
You may like
-
 183133-96-2 Cabazitaxel 98%View Details
183133-96-2 Cabazitaxel 98%View Details
183133-96-2 -
 183133-96-2 98%View Details
183133-96-2 98%View Details
183133-96-2 -
 Cabazitaxel 98%View Details
Cabazitaxel 98%View Details
183133-96-2 / 1402820-62-5 -
 Cabazitaxel 183133-96-2 / 1402820-62-5 99%View Details
Cabazitaxel 183133-96-2 / 1402820-62-5 99%View Details
183133-96-2 / 1402820-62-5 -
 183133-96-2 / 1402820-62-5 Cabazitaxel 98%View Details
183133-96-2 / 1402820-62-5 Cabazitaxel 98%View Details
183133-96-2 / 1402820-62-5 -
 183133-96-2 / 1402820-62-5 98%View Details
183133-96-2 / 1402820-62-5 98%View Details
183133-96-2 / 1402820-62-5 -
 Cabazitaxel 98%View Details
Cabazitaxel 98%View Details
183133-96-2 / 1402820-62-5 -
 Cabazitaxel 183133-96-2 / 1402820-62-5 98%View Details
Cabazitaxel 183133-96-2 / 1402820-62-5 98%View Details
183133-96-2 / 1402820-62-5