Aluminum sulfate
Synonym(s):Aluminum sulfate hydrate;Aluminum sulfate octahydrate
- CAS NO.:10043-01-3
- Empirical Formula: Al2O12S3
- Molecular Weight: 342.15
- MDL number: MFCD00003423
- EINECS: 233-135-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-05-21 21:51:03
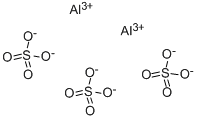
What is Aluminum sulfate?
Absorption
The degree of aluminum absorption depends on a number of factors, such as the aluminum salt ingested, pH (for aluminum speciation and solubility), bioavailability, as well as dietary conditions .
These facts should be taken into consideration during tissue dosimetry and response assessment to aluminum sulfate. It can be concluded that the use of currently available animal studies to develop a guideline value is inappropriate at this time due to the above specific toxicokinetic/dynamic factors that may affect results .
Toxicity
Aluminum Sulfate, Hydrated (ACS & FCC): ORAL (LD50):
Acute: >9000 mg/kg in the mouse . >9000 mg/kg in the rat .
Acute Toxicity
There is little indication that aluminum is acutely toxic by oral exposure despite it is widely found in foods, drinking water, and many antacid preparations . In 1988, a population of about 20 000 citizens of Camelford, England, was exposed to increased levels of aluminum for 5 days. The aluminum was accidentally ingested by the population from a water supply facility using aluminum sulfate for water treatment .
Some adverse effects observed were nausea, vomiting, diarrhea, mouth ulcers, skin ulcers, skin rashes, and arthritis-type pain were observed. It was concluded in one study that the adverse effects of aluminum sulfate were primarily mild and transient. No long-lasting effects on health could be attributed to the exposures from aluminum in the drinking water during this period .
Chronic Toxicity
In humans, excess exposure to aluminum via dialysis water (aluminum sulfate) is a known etiological factor in several pathological conditions in patients treated with hemodialysis. Clinical symptoms and signs of aluminum toxicity include hypercalcemia, anemia, vitamin D refractory osteodystrophy, and a dialysis encephalopathy. Bone pain, pathological fractures, and proximal myopathy may occur. Aluminum has also been suggested as an etiology of several neurodegenerative diseases such as Alzheimer senile and pre-senile dementia, as well amyotrophic sclerosis. Despite this, the most recent investigations have failed to confirm this hypothesis. A study in man has verified a number of possible deleterious interactions of aluminum salts with phosphorous metabolism, especially in long-term ingestion of aluminum-containing antacids .
It has been suggested that aluminum exposure is a risk factor for the development or acceleration of onset of Alzheimer disease (AD) in humans. The world health organization has completed a meta-analysis of 20 epidemiological studies done to test the hypothesis that aluminum in drinking-water is a risk factor for Alzheimer disease. Six studies on populations in Norway were considered of sufficiently high quality to meet the general criteria for exposure and outcome assessment and the adjustment for at least some confounding variables .
Of six studies that examined the relationship between aluminum in drinking- water and dementia, three found a positive relationship, but three did not. However, each of the studies had significant deficiencies in the study design (e.g. ecological exposure assessment; failure to consider aluminum exposure from all sources and to control for important confounders, such as education, socioeconomic status, and family history; the use of surrogate outcome measures for AD; and selection bias) .
In general, the relative risks determined were less than 2, with large confidence intervals, when the total aluminum concentration in drinking-water was 0.1 mg/L or higher. Due to the pathogenesis of AD and knowledge obtained from studies, it was concluded that the present epidemiological evidence does not support a causal association between AD and aluminum in drinking-water .
In addition to the epidemiological studies that examined the relationship between AD and aluminum in drinking-water, two studies studied cognitive dysfunction in elderly populations in relation to the levels of aluminum in drinking water. The results proved conflicting. A study of 800 male subjects, age 80-89, drinking water containing aluminum concentrations up to 98 μg/L found no relationship. The second study used “any evidence of mental impairment” as an outcome measure and found a relative risk of 1.72 at aluminum drinking-water concentrations above 85 μg/L in 250 males. Such data are insufficient to show that aluminum is a cause of cognitive impairment in the elderly .
Note on possible risk of breast cancer
Widespread concern has been raised regarding the exposure to aluminum in deodorant/antiperspirant products, with inconclusive results , , , . Results from a more recent case-control study suggest an association between underarm cosmetic use and aluminum concentration in breast tissue and breast cancer. The observed association of underarm cosmetic use with breast cancer was, however, limited to women who report using the products multiple times a day before age of 30 .
The Uses of Aluminum sulfate
Aluminum sulfate acts as a flocculating agent in the purification of drinking water, and in waste water treatment plants. It acts as a mordant in dyeing, printing, textiles and also used in paper manufacturing. It is a waterproofing agent and accelerator in concrete. Further, it is used as a foaming agent in fire fighting foam, photographic film and in photochemicals. It is also used in styptic pencils, pain relief and in dentistry for gingival retraction cords.
Indications
Solutions containing 5 to 10% aluminum sulfate have been used as local applications to ulcers and to arrest foul discharges from mucous surfaces. Aluminum sulfate is also used in the preparation of aluminum acetate ear drops . It is often purchased over the counter and is available in solid stick or powder form for minor cuts and abrasions after shaving , . Aluminum sulfate is also used as an adjuvant in vaccines .
Background
Aluminum (Al), also spelled aluminum, chemical element, a lightweight, silvery-white metal of main Group 13 (IIIa, or boron group) of the periodic table .
It is a chemical agent used in water purification, the pH regulation of garden soil, and other commercial or industrial applications. Medically, it is primarily used as a coagulating agent in minor cuts and abrasions as well as deodorant .
Aluminum (Al) is ubiquitous and represents the third most common element in the Earth’s crust. It most commonly exists in a combined state with various other elements. Al is found in materials used in the pharmaceutical industry, and in manufactured foodstuffs, cosmetics, and tap water. By overcoming the body barriers, Al may infiltrate into the blood and lead to toxic effects in liver, bone and the central nervous system .
Pharmacokinetics
Aluminum sulfate may be used as a deodorant, as well as an astringent . Aluminum sulfate is also known as an astringent. Astringents are substances that cause contraction or shrinkage of tissues and that dry up secretion .
Used as a post-shaving treatment, it can eliminate bleeding from superficial wounds , .
It has also shown in vitro anti-microbial activity .
Metabolism
Not Available
Properties of Aluminum sulfate
| Melting point: | 770 °C (dec.) (lit.) |
| Boiling point: | 759.71°C (estimate) |
| Density | 2.71 g/mL at 25 °C (lit.) |
| solubility | Soluble in cold water, freely soluble in hot water, practically insoluble in ethanol (96 per cent). |
| form | Powder and/or Chunks |
| color | White |
| Water Solubility | soluble |
| Sensitive | Hygroscopic |
Safety information for Aluminum sulfate
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05 |
| GHS Hazard Statements |
H290:Corrosive to Metals H318:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P234:Keep only in original container. P280:Wear protective gloves/protective clothing/eye protection/face protection. P390:Absorb spillage to prevent material damage. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Aluminum sulfate
Abamectin manufacturer
sheetalchemicals
UNILOSA INTERNATINAL PRIVATE LIMITED
JSK Chemicals
Ultra Chemical Works
Kronox Lab Sciences Pvt Ltd
Vats International
Agilenobel
New Products
3-N-BOC-(S)-AMINO BUTYRONITRILE 4-Piperidinopiperidine N-Benzyl-3-hydroxypiperidine 2-Methyl-4-nitrobenzoic acid 2-(4-bromophenyl)-2-methylpropanoic acid 4-Acetyl-2-methylbenzoicacid Acetyl-meldrum's acid Ethyl-4-Pyrazole carboxylate 2,6-Pyridinedimethanol 5,7-Dichloro-3H-Imidazo[4,5-B]Pyridine 5-Bromo-2-Methoxy-4-Methyl-3-Nitropyridine 2-Fluoro-5-Iodopyridine 2-Fluoro-5-Methylpyridine 2-Chloro-3-Bromo-5-Amiopyridine METHYL-4-(BUTYRYLAMINO)3-METHYL-5-NITROBENZOATE TRANS-CYCLOBUTANE-1,2- DICARBOXYLIC ACID 5-Nitro indazole R-(-)-5-(2-AMINO-PROPYL)-2-METHOXY-BENZENESULFONAMIDE 1,3-cyclohexanedione 4-Aminophenaethylalchol (S)-(+)-4-BENZYL-2-OXAZOLIDINONE 3-NITRO-5-ACETYL IMINODIBENZYL 4-FLUORO PHENYL MAGNESIUM BROMIDE 1.0 M IN THF 1-HYDROXY-4-METHYL6-(2,4,4-TRI METHYL PHENYL)-2-PYRIDONE MONO ETHANOL AMINE(PIROCTONE OLAMINE)Related products of tetrahydrofuran




You may like
-
 ALUMINIUM SULPHATE 99%View Details
ALUMINIUM SULPHATE 99%View Details -
 Aluminium Sulphate 99%View Details
Aluminium Sulphate 99%View Details
10043-01-3 -
 Aluminum sulfate, 98% 99%View Details
Aluminum sulfate, 98% 99%View Details -
 Aluminium Sulphate 99%View Details
Aluminium Sulphate 99%View Details -
 Aluminum sulfate 98%View Details
Aluminum sulfate 98%View Details -
 Aluminum sulfate 10043-01-3 98%View Details
Aluminum sulfate 10043-01-3 98%View Details
10043-01-3 -
 10043-01-3 Aluminum sulfate 98%View Details
10043-01-3 Aluminum sulfate 98%View Details
10043-01-3 -
 Aluminum sulfate 10043-01-3 98%View Details
Aluminum sulfate 10043-01-3 98%View Details
10043-01-3