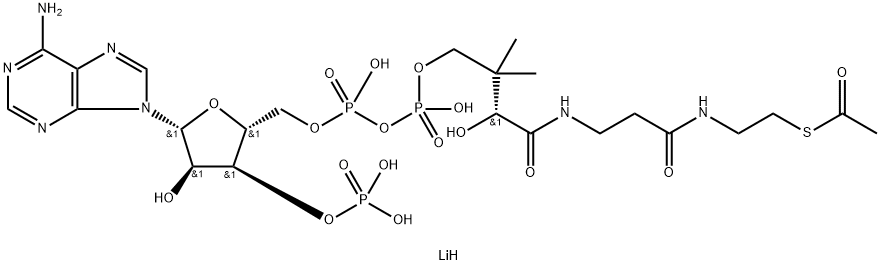
Acetyl coenzyme A sodium salt
Synonym(s):acetyl-coa
- CAS NO.:102029-73-2
- Empirical Formula: C23H38N7O17P3S.3Na
- Molecular Weight: 809.57
- MDL number: MFCD00078858
- SAFETY DATA SHEET (SDS)
- Update Date: 2026-06-04 18:03:49
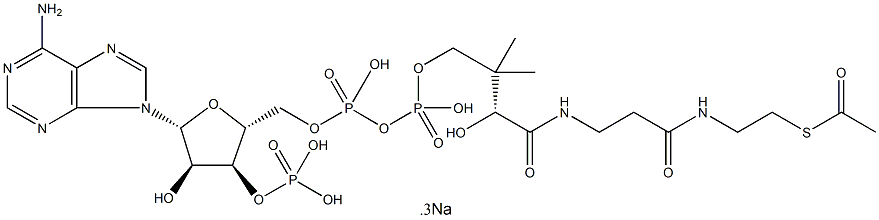
What is Acetyl coenzyme A sodium salt?
Description
Acetyl-coenzyme A (acetyl-
The Uses of Acetyl coenzyme A sodium salt
Acetyl-Coenzyme A is used to assay CAT enzyme activity in cell extracts using radioisotopes. CAT enzyme activity in cell extracts catalyzes the transfer of acetyl groups from acetylcoenzyme A to chloramphenicol. A number of assays have been developed to measure CAT activity in cell extracts. Acetyl-Coenzyme A has also been used to determine citrate synthase activity.
What are the applications of Application
Acetyl coenzyme A sodium salt is an essential cofactor, metabolic intermediate, and carrier of acyl groups.
General Description
Acetyl-Coenzyme A, trilithium salt
Biochem/physiol Actions
Acetyl-Coenzyme A (Ac-CoA) is the end product of glycolysis and takes part in the Ac-CoA pathway, which is a metabolic pathway for carbon compounds. Ac-CoA is important in cholesterol synthesis. It also takes part in fatty acid biosynthesis and catabolism of polyamines like spermine and spermidine. A low level of Ac-CoA leads to a loss in glial cell and neuronal function. Ketone bodies and triglycerides give rise to Ac-CoA on hydrolysis and this indirectly leads to increased histone acetylation.
Metabolism
Acetyl coenzyme A, the precursor of the acetate-malonare pathway, is a metabolite of extreme importance in both primary and secondary metabolism. In every living organism, there exists a metabolie pool of acetyl CoA which is continually replenished and depleted. Glycolysis and the catabolism of fatty acids and amino acids produce acetyl CoA, while this compound is the precursor of a host of primary and secondary metabolites, including fatty acids, terpenoids, steroids, polyketides, aromatic compounds and acetyl esters and amides. The conversion of acetyl CoA to citrate and other tricarboxylic acids leads to the formation of the amino acids and their products, such as the nucleic acids and alkaloids.
Acetyl CoA is also central to the catabolism of glucose and the fatty acids, which, through the tricarboxylic acid cycle, are the main reaction sequences producing the energy necessary for metabolie processes.
In plants, an enzyme, acetyl CoA synthetase, catalyses the formation of the thiol ester from acetate.
Properties of Acetyl coenzyme A sodium salt
| storage temp. | -20°C |
| solubility | H2O: 100 mg/mL |
| form | powder |
| color | White to off-white |
Safety information for Acetyl coenzyme A sodium salt
Computed Descriptors for Acetyl coenzyme A sodium salt
| InChIKey | ZSLZBFCDCINBPY-KMYLAXNMSA-N |
New Products
3-AMINO-3-(2-FLUORO-PHENYL)-PROPIONIC ACID Pentadecanoic acid Hendecanoic acid 2-AMINO-3-METHYLQUINOLINE HYDROCHLORIDE 3-Hydroxypropionitrile DL-3-Amino-3-(2-methoxyphenyl)propionic acid 2-Bromo-5-Iodopyridine 2,3-Diamino-5-Chloropyridine 2-Amino-3-Hydroxypyridine 2,6-Diamino Pyridine 4-Amino-2-Chloropyridine 2-Hydroxy-4-Picoline 4-(4-(Dimethylamino)-1-(4-fluorophenyl)-1-hydroxybutyl)-3-(hydroxymethyl)benzonitrile 2-Hydroxy-5-methoxybenzoic acid 2-Cyanopyrimidine Xanthene-9-carboxylic acid 4,4-Diethoxybutanenitrile Methyl 4-amino-2,3-difluoro-5-nitrobenzoate 5-fluoro-1,3-benzodioxole 5-Phenyl-[1,3,4]-thiadiazol-2-amine 2-Chloro-6-nitro benzothiazole 4-Chloro-2-methyl quinoline 2-Amino-4-phenyl-thiazole 2-(Chloromethyl) quinazolin-4(3H)-oneRelated products of tetrahydrofuran


You may like
-
 Acetyl coenzyme A trisodium salt CAS 102029-73-2View Details
Acetyl coenzyme A trisodium salt CAS 102029-73-2View Details
102029-73-2 -
 Acetyl-Coenzyme A CASView Details
Acetyl-Coenzyme A CASView Details -
 2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
2-Bromo-4-nitropyridine-N-oxide 52092-43-0 98%View Details
52092-43-0 -
 18979-61-8 98%View Details
18979-61-8 98%View Details
18979-61-8 -
 4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
4, 4'-Ditolylamine (or) 4,4-Dimethyl Diphenylamine 620-93-9 98%View Details
620-93-9 -
 2-Bromo-5-Chloropyridine 98%View Details
2-Bromo-5-Chloropyridine 98%View Details
40473-01-6 -
 2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
2-(2,4-Diaminophenoxy)ethanol Dihydrochloride 98%View Details
66422-95-5 -
 Eperisone Hydrochloride APIView Details
Eperisone Hydrochloride APIView Details
56839-43-1
