83-88-5
CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Yellow to orange-yellow crystalline powder, with slight odour |
|---|---|
| Color/Form | Fine orange-yellow needles from 2N acetic acid, alcohol, water, or pyridine ... three different crystal forms |
| Odor | Slight odor |
| Taste | Bitter |
| Melting Point | 280 dec °C |
| Solubility | 84.7 mg/L (at 25 °C) |
| LogP | -1.46 |
| LogS | -3.68 |
| Stability/Shelf Life | Crystalline riboflavin shows no evidence of decomposition under ordinary conditions, but protection from light is advisable |
| Optical Rotation | [α]D/20 between – 115° and – 140° in a 0,05 N sodium hydroxide solution |
| Decomposition | When heated to decomposition it emits toxic fumes of /nitric oxide/. |
| pH | pH of saturated aqueous solution: about 6 |
| Dissociation Constants | 10.2 |
| Collision Cross Section | 188.3 Ų [M+H]+ [CCS Type: DT, Method: single field calibrated with Agilent tune mix (Agilent)] |
| Other Experimental Properties | Visible or ultraviolet radiation of alkaline solutions causes the formation of lumiflavine ... whereas irradiation of acid or neutral solutions gives rise to the production of lumichrome ... togther with varying amounts of lumiflavine |
| Chemical Classes | Biological Agents -> Vitamins and Derivatives |
SAFETY INFORMATION
| Signal word | Danger |
|---|---|
| Pictogram(s) |
 Flame Flammables GHS02  Skull and Crossbones Acute Toxicity GHS06  Health Hazard GHS08 |
| GHS Hazard Statements |
H226:Flammable liquids H370:Specific target organ toxicity, single exposure |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P280:Wear protective gloves/protective clothing/eye protection/face protection. |
COMPUTED DESCRIPTORS
| Molecular Weight | 376.4 g/mol |
|---|---|
| XLogP3 | -1.5 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 5 |
| Exact Mass | 376.13828437 g/mol |
| Monoisotopic Mass | 376.13828437 g/mol |
| Topological Polar Surface Area | 155 Ų |
| Heavy Atom Count | 27 |
| Formal Charge | 0 |
| Complexity | 680 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 3 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
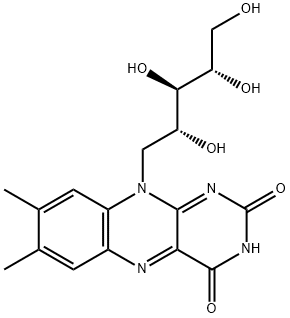
Riboflavin is d-Ribitol in which the hydroxy group at position 5 is substituted by a 7,8-dimethyl-2,4-dioxo-3,4-dihydrobenzo[g]pteridin-10(2H)-yl moiety. It is a nutritional factor found in milk, eggs, malted barley, liver, kidney, heart, and leafy vegetables, but the richest natural source is yeast. The free form occurs only in the retina of the eye, in whey, and in urine; its principal forms in tissues and cells are as flavin mononucleotide and flavin-adenine dinucleotide. It has a role as a photosensitizing agent, a food colouring, an Escherichia coli metabolite, a mouse metabolite, a cofactor, a plant metabolite, a human urinary metabolite, an anti-inflammatory agent, a fundamental metabolite and an antioxidant. It is a vitamin B2 and a flavin. It is a conjugate acid of a riboflavin(1-).