133-32-4
Product Name:
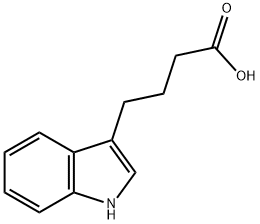
Indole-3-butyric acid
Formula:
C12H13NO2
Synonyms:
4-(3-Indolyl)butanoic acid;4-(3-Indolyl)butyric acid;IBA
Inquiry
CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | White or slightly yellow solid; [Merck Index] Off-white powder; [MSDSonline] |
|---|---|
| Color/Form | White to slightly yellow crystals |
| Odor | Essentially odorless |
| Melting Point | 124.5 °C |
| Solubility | 250 mg/L (at 20 °C) |
| Vapor Pressure | 0.00000117 [mmHg] |
| LogP | 2.3 |
| Henry's Law Constant | Henry's Law constant: 1.3X10-11 atm cu m/mol@ 25 °C /Estimated/ |
| Stability/Shelf Life | Very stable in neutral, acidic and alkaline media. |
| Dissociation Constants | pKa = 4.7 /Estimated/ |
| Kovats Retention Index | 1885 |
| Other Experimental Properties | Hydroxyl radical reaction rate constant: 2.0X10-10 cu cm/molecule-sec @ 25 °C /Estimated/ |
| Chemical Classes | Pesticides -> Plant Growth Regulators |
SAFETY INFORMATION
| Signal word | Danger |
|---|---|
| Pictogram(s) |
 Skull and Crossbones Acute Toxicity GHS06 |
| GHS Hazard Statements |
H301:Acute toxicity,oral |
| Precautionary Statement Codes |
P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P270:Do not eat, drink or smoke when using this product. P301+P310:IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician. P405:Store locked up. P501:Dispose of contents/container to..… |
COMPUTED DESCRIPTORS
| Molecular Weight | 203.24 g/mol |
|---|---|
| XLogP3 | 2.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 4 |
| Exact Mass | 203.094628657 g/mol |
| Monoisotopic Mass | 203.094628657 g/mol |
| Topological Polar Surface Area | 53.1 Ų |
| Heavy Atom Count | 15 |
| Formal Charge | 0 |
| Complexity | 230 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Indole-3-butyric acid is a indol-3-yl carboxylic acid that is butanoic acid carrying a 1H-indol-3-yl substituent at position 1. It has a role as a plant hormone, a plant metabolite and an auxin. It is functionally related to a butyric acid. It is a conjugate acid of an indole-3-butyrate.