

Silica, fumed
| Price | USD1.00 |
| Packge | 1KG |
- Min. Order:1KG
- Supply Ability:100kg
- Time:2019-12-23
Product Details
- Product NameSilica, fumed
- CAS No.112945-52-5
- EINECS No.231-545-4
- MFO2Si
- MW60.08
- Appearancepowder
- density 2.3 lb/cu.ft at 25 °C (bulk density)(lit.)
- Melting point >1600°C
▼
▲
Product Name:
Synonyms:
SILICA, FUMED;SILICA, FUMED;acticel;SILICA GEL 7G;SILICA GEL 8-20 MESH;SILICA GEL 12-28 MESH;SILICA GEL 100;SILICA GEL 60
CAS:
MF:
O2Si
MW:
60.08
EINECS:
231-545-4
Product Categories:
Mol File:
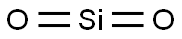
▼
▲
Silica, fumed Chemical Properties
▼
▲
Melting point
>1600°C
density
2.3 lb/cu.ft at 25 °C (bulk density)(lit.)
refractive index
n20/D 1.46(lit.)
solubility
Practically insoluble in organic solvents, water, and acids, except hydrofluoric acid; soluble in hot solutions of alkali hydroxide. Forms a colloidal dispersion with water. For Aerosil, solubility in water is 150 mg/L at 258℃ (pH 7).
form
powder
CAS DataBase Reference
EPA Substance Registry System
▼
▲
Safety Information
▼
▲
▼
▲
Silica, fumed Usage And Synthesis
▼
▲
Chemical Properties
Amorphous silica, the noncrystalline form of SiO2, is a transparent to gray, odorless, amorphous powder
Chemical Properties
Colloidal silicon dioxide is a submicroscopic fumed silica with a particle size of about 15 nm. It is a light, loose, bluish-white-colored, odorless, tasteless, amorphous powder.
Production Methods
Colloidal silicon dioxide is prepared by the flame hydrolysis of chlorosilanes, such as silicon tetrachloride, at 18008℃ using a hydrogen–oxygen flame. Rapid cooling from the molten state during manufacture causes the product to remain amorphous.
Pharmaceutical Applications
Colloidal silicon dioxide is widely used in pharmaceuticals, cosmetics, and food products. Its small particle size and large specific surface area give it desirable flow characteristics that are exploited to improve the flow properties of dry powders in a number of processes such as tableting and capsule filling.
Colloidal silicon dioxide is also used to stabilize emulsions and as a thixotropic thickening and suspending agent in gels and semisolid preparations. With other ingredients of similar refractive index, transparent gels may be formed. The degree of viscosity increase depends on the polarity of the liquid (polar liquids generally require a greater concentration of colloidal silicon dioxide than nonpolar liquids). Viscosity is largely independent of temperature. However, changes to the pH of a system may affect the viscosity1.
In aerosols, other than those for inhalation, colloidal silicon dioxide is used to promote particulate suspension, eliminate hard settling, and minimize the clogging of spray nozzles. Colloidal silicon dioxide is also used as a tablet disintegrant and as an adsorbent dispersing agent for liquids in powders. Colloidal silicon dioxide is frequently added to suppository formulations containing lipophilic excipients to increase viscosity, prevent sedimentation during molding, and decrease the release rate. Colloidal silicon dioxide is also used as an adsorbent during the preparation of wax microspheres; as a thickening agent for topical preparations; and has been used to aid the freeze-drying of nanocapsules and nanosphere suspensions.
Colloidal silicon dioxide is also used to stabilize emulsions and as a thixotropic thickening and suspending agent in gels and semisolid preparations. With other ingredients of similar refractive index, transparent gels may be formed. The degree of viscosity increase depends on the polarity of the liquid (polar liquids generally require a greater concentration of colloidal silicon dioxide than nonpolar liquids). Viscosity is largely independent of temperature. However, changes to the pH of a system may affect the viscosity1.
In aerosols, other than those for inhalation, colloidal silicon dioxide is used to promote particulate suspension, eliminate hard settling, and minimize the clogging of spray nozzles. Colloidal silicon dioxide is also used as a tablet disintegrant and as an adsorbent dispersing agent for liquids in powders. Colloidal silicon dioxide is frequently added to suppository formulations containing lipophilic excipients to increase viscosity, prevent sedimentation during molding, and decrease the release rate. Colloidal silicon dioxide is also used as an adsorbent during the preparation of wax microspheres; as a thickening agent for topical preparations; and has been used to aid the freeze-drying of nanocapsules and nanosphere suspensions.
Safety Profile
Poison by intraperitoneal, intravenous, and intratracheal routes. Moderately toxic by ingestion. An inhalation hazard. Much less toxic than crystalhe forms. Questionable carcinogen with experimental carcinogenic data. Mutation data reported. Does not cause silicosis. See also other silica entries
Safety
Colloidal silicon dioxide is widely used in oral and topical pharmaceutical products and is generally regarded as an essentially nontoxic and nonirritant excipient. However, intraperitoneal and subcutaneous injection may produce local tissue reactions and/or granulomas. Colloidal silicon dioxide should therefore not be administered parenterally.
LD50 (rat, IV): 0.015 g/kg
LD50 (rat, oral): 3.16 g/kg
LD50 (rat, IV): 0.015 g/kg
LD50 (rat, oral): 3.16 g/kg
Potential Exposure
Amorphous fumed silica is used as a mineral, natural or synthetic fiber. A potential danger to those involved in the production and handling of fumed silica for paint pigments or catalysts. Diatomaceous earth is used in clarifying liquids, in manufacture of fire brick and heat insulators; used as a filtering agent; as a filler in construction materials; pesticides, paints, and varnishes. A potential danger to those involved in mining of diatomaceous earth or fabrication of products there from.
storage
Colloidal silicon dioxide is hygroscopic but adsorbs large quantities of water without liquefying. When used in aqueous systems at a pH 0–7.5, colloidal silicon dioxide is effective in increasing the viscosity of a system. However, at a pH greater than 7.5 the viscosityincreasing properties of colloidal silicon dioxide are reduced; and at a pH greater than 10.7 this ability is lost entirely since the silicon dioxide dissolves to form silicates. Colloidal silicon dioxide powder should be stored in a well-closed container.
Purification Methods
Purification of silica for high technology applications uses isopiestic vapour distillation from concentrated volatile acids and is absorbed in high purity water. The impurities remain behind. Preliminary cleaning to remove surface contaminants uses dip etching in HF or a mixture of HCl, H2O2 and deionised water [Phelan & Powell Analyst 109 1299 1984].
Incompatibilities
Silica, amorphous is a noncombustible solid. Generally unreactive chemically. Incompatible with fluorine, oxygen difluoride, chlorine trifluoride. Soluble in molten alkalis and reacts with most metallic oxides at high temperature.
Incompatibilities
Incompatible with diethylstilbestrol preparations.
Waste Disposal
Sanitary landfill.
Regulatory Status
GRAS listed. Included in the FDA Inactive Ingredients Database (oral capsules, suspensions, and tablets; transdermal, rectal, and vaginal preparations). Also approved by the FDA as a food additive and for food contact. Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Nonmedicinal Ingredients.
Company Profile Introduction
Established in 2014,Career Henan Chemical Co. is a manufacturerspecializing in the sale of fine chemicals.
Mainly deals in the sales of:
Pharmaceutical intermediates
OLED intermediates:
Pharmaceutical intermediates;
OLED intermediates;
Recommended supplier
-
VIP1年
- 20 Microns Limited
- Silica,Amorphous fumed 98%
- Inquiry
- 2024-02-20
-
VIP1年
- Merck Ltd
- 10279-57-9 / 12508-66-6 98%
- Inquiry
- 2024-01-17
-
VIP1年
- Agilenobel
- 112945-52-5 / 69012-64-2 Silica,Amorphous fumed 98%
- Inquiry
- 2024-01-16
- Since:2014-12-17
- Address: Room 702, Floor 7, Building 10, National University Science Park, High-Tech Zone, Zhengzhou City, H
INQUIRY
