

Magnesium carbonate
| Price | USD1.00 |
| Packge | 1KG |
- Min. Order:1KG
- Supply Ability:100kg
- Time:2019-12-20
Product Details
- Product NameMagnesium carbonate
- CAS No.546-93-0
- EINECS No.208-915-9
- MFCMgO3
- MW84.31
- InChIKeyZLNQQNXFFQJAID-UHFFFAOYSA-L
- AppearanceSolidWhite
- Melting point 990°C
- density 3.050
- Water Solubility g MgCO3/100g solution at CO2 pressure, kPa, 18°C: 3.5 (203), 4.28 (405), 5.90 (1010), 7.49 (1820), 7.49 (5670); at 0°C 8.58 (3445), at 60°C 5.56 (3445); soluble acids; insoluble alcohol [HAW93] [KIR81]
▼
▲
▼
▲
Product Name:
Synonyms:
carbonicacid,magnesiumsalt;Carbonicacid,magnesiumsalt(1:1);ci77713;DCI Light Magnesium Carbonate;dcilightmagnesiumcarbonate;barringtonite;C.I. 77713;c.i.77713
CAS:
MF:
CMgO3
MW:
84.31
EINECS:
208-915-9
Product Categories:
Mol File:
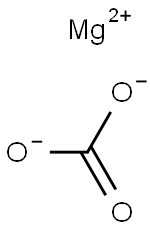
▼
▲
Magnesium carbonate Chemical Properties
▼
▲
Melting point
990°C
density
3.050
solubility
Practically insoluble in water. It dissolves in dilute acids with effervescence.
form
Solid
color
White
Water Solubility
g MgCO3/100g solution at CO2 pressure, kPa, 18°C: 3.5 (203), 4.28 (405), 5.90 (1010), 7.49 (1820), 7.49 (5670); at 0°C 8.58 (3445), at 60°C 5.56 (3445); soluble acids; insoluble alcohol [HAW93] [KIR81]
CAS DataBase Reference
NIST Chemistry Reference
EPA Substance Registry System
▼
▲
Safety Information
▼
▲
Safety Statements
RTECS
OM2470000
HS Code
28369910
Hazardous Substances Data
▼
▲
Provider
Language
English
▼
▲
Magnesium carbonate Usage And Synthesis
▼
▲
Compound Mineral Formula CAS No.
anhydrous salt magnesite MgCO3 [13717-00-5]
dihydrate barringtonite MgCO3•2H2O [5145-48-2]
trihydrate nesquehonite MgCO3•3H2O [14457-83-1]
pentahydrate lansfordite MgCO3•5H2O [61042-72-6]
basic carbonate artinite MgCO3•Mg(OH)2•3H2O [12143-96-3]
basic carbonate hydromagnestite 4MgCO3•Mg(OH)2•4H2O [12072-90-1]
basic carbonate dypingite 4MgCO3•Mg(OH)2•5H2O [12544-02-4]
basic carbonate --- 4MgCO3•Mg(OH)2•8H2O [75300-49-1]
anhydrous salt magnesite MgCO3 [13717-00-5]
dihydrate barringtonite MgCO3•2H2O [5145-48-2]
trihydrate nesquehonite MgCO3•3H2O [14457-83-1]
pentahydrate lansfordite MgCO3•5H2O [61042-72-6]
basic carbonate artinite MgCO3•Mg(OH)2•3H2O [12143-96-3]
basic carbonate hydromagnestite 4MgCO3•Mg(OH)2•4H2O [12072-90-1]
basic carbonate dypingite 4MgCO3•Mg(OH)2•5H2O [12544-02-4]
basic carbonate --- 4MgCO3•Mg(OH)2•8H2O [75300-49-1]
Magnesium carbonate occurs in nature in several minerals as hydrated, basic and double salts, as shown above. The two principal minerals are magnesite, MgCO3 and dolomite, a double salt, CaCO3•MgCO3. Both minerals are used as source materials in the production of magnesium metal. Also, they are calcined to produce basic refractory bricks. Other applications of magnesium carbonate are in flooring, fireproofing and fire-extinguishing compositions; as a filler material and smoke suppressant in plastics; as a reinforcing agent in neoprene rubber; as a drying agent and for color retention in foods; in cos-metics; in dusting powder; and in toothpaste. The high purity magnesium carbonate is used as an antacid in medicine; and as an additive to table salt. Another important application of magnesium carbonate is as a starting material in producing a number of magnesium compounds.
Description
Magnesium carbonate is obtained mainly by mining the natural mineral magnesite. The trihydrate salt, MgCO3·3H2O, is prepared by mixing solutions of magnesium and carbonate ions in the presence of carbon dioxide.
Chemical Properties
White or almost white powder.
Chemical Properties
Magnesium carbonate occurs as light, white-colored friable masses or as a bulky, white-colored powder. It has a slightly earthy taste and is odorless but, since it has a high absorptive ability, magnesium carbonate can absorb odors.
The USP 32 describes magnesium carbonate as either a basic hydrated magnesium carbonate or a normal hydrated magnesium carbonate. However, the PhEur describes magnesium carbonate as being a hydrated basic magnesium carbonate in two separate monographs: heavy magnesium carbonate and light magnesium carbonate. The molecular formulas for heavy magnesium carbonate and light magnesium carbonate vary, but heavy magnesium carbonate may generally be regarded as the tetrahydrate [(MgCO3)3·Mg(OH)2·4H2O], while light magnesium carbonate may be regarded as the trihydrate [(MgCO3)3· Mg(OH)2·3H2O].
The molecular weights of the heavy and light forms of magnesium carbonate are 383.32 and 365.30, respectively.
The USP 32 describes magnesium carbonate as either a basic hydrated magnesium carbonate or a normal hydrated magnesium carbonate. However, the PhEur describes magnesium carbonate as being a hydrated basic magnesium carbonate in two separate monographs: heavy magnesium carbonate and light magnesium carbonate. The molecular formulas for heavy magnesium carbonate and light magnesium carbonate vary, but heavy magnesium carbonate may generally be regarded as the tetrahydrate [(MgCO3)3·Mg(OH)2·4H2O], while light magnesium carbonate may be regarded as the trihydrate [(MgCO3)3· Mg(OH)2·3H2O].
The molecular weights of the heavy and light forms of magnesium carbonate are 383.32 and 365.30, respectively.
Uses
Used in Pharmaceutical units as a Magnesium Salt, Heat Insulator and Refractor, Antacid. Also used in Cosmetics, inks, glass, drying agent, color retention agent and etc.
Uses
Some applications of magnesiumcarbonate are uses in flooring, fireproofing and fire-extinguishing compositions; as a filler material and smoke suppressant in plastics; as a reinforcing agent in neoprene rubber; as a drying agent and for color retention in foods; in cosmetics. Magnesium carbonate is used as an antacid in medicine and as an additive to table salt. Another important application of magnesium carbonate is as a starting material in producing a number of magnesium compounds.
Because of its water-insoluble, hygroscopic properties, MgCO3 was first added to salt in 1911 to make the salt flow more freely. Magnesium carbonate, most often referred to as “chalk”, is used as a drying agent for hands in gymnastics, weight lifting and rock climbing. Magnesium carbonate is also used in taxidermy for whitening skulls. It can be mixed with hydrogen peroxide to create a paste, which is then spread on the skull to give it awhite finish. Basic magnesium carbonate is used as a clay in face masks and it has mild astringent properties and helps to smooth and soften skin. It is recommended for use on normal to dry skins.
Because of its water-insoluble, hygroscopic properties, MgCO3 was first added to salt in 1911 to make the salt flow more freely. Magnesium carbonate, most often referred to as “chalk”, is used as a drying agent for hands in gymnastics, weight lifting and rock climbing. Magnesium carbonate is also used in taxidermy for whitening skulls. It can be mixed with hydrogen peroxide to create a paste, which is then spread on the skull to give it awhite finish. Basic magnesium carbonate is used as a clay in face masks and it has mild astringent properties and helps to smooth and soften skin. It is recommended for use on normal to dry skins.
Production Methods
Depending upon the manufacturing process used, the composition of the magnesium carbonate obtained may vary from normal hydrated magnesium carbonate to basic hydrated magnesium carbonate.
Light magnesium carbonate may be manufactured by saturating an aqueous suspension of dolomite, CaMg(CO3)2, with carbon dioxide under pressure. On increase of the temperature, calcium carbonate precipitates almost entirely. The filtered solution is then heated to boiling; the magnesium bicarbonate in the solution loses precipitates.
Heavy magnesium carbonate may be manufactured by mixing a hot concentrated solution of magnesium chloride or magnesium sulfate with a solution of sodium carbonate. The heavy magnesium carbonate may be either precipitated to produce a granular material or spray-dried. Varying the temperature of the reaction solutions produces heavy magnesium carbonate with differing physical properties: e.g. material with a higher specific surface area is produced at a lower reaction temperature. Low processing temperature provided the largest surface area, which produced optimum granules or spray-dried powder.If dilute magnesium chloride or magnesium sulfate solutions are used for the reaction, a less dense material is produced.
Magnesium carbonates in varying states of hydration are also found as minerals in nature.
Light magnesium carbonate may be manufactured by saturating an aqueous suspension of dolomite, CaMg(CO3)2, with carbon dioxide under pressure. On increase of the temperature, calcium carbonate precipitates almost entirely. The filtered solution is then heated to boiling; the magnesium bicarbonate in the solution loses precipitates.
Heavy magnesium carbonate may be manufactured by mixing a hot concentrated solution of magnesium chloride or magnesium sulfate with a solution of sodium carbonate. The heavy magnesium carbonate may be either precipitated to produce a granular material or spray-dried. Varying the temperature of the reaction solutions produces heavy magnesium carbonate with differing physical properties: e.g. material with a higher specific surface area is produced at a lower reaction temperature. Low processing temperature provided the largest surface area, which produced optimum granules or spray-dried powder.If dilute magnesium chloride or magnesium sulfate solutions are used for the reaction, a less dense material is produced.
Magnesium carbonates in varying states of hydration are also found as minerals in nature.
Definition
A whitecompound, MgCO3, existing in anhydrousand hydrated forms. The anhydrousmaterial (trigonal; r.d.2.96) is found in the mineral magnesite.There is also a trihydrate,MgCO3.3H2O (rhombic; r.d. 1.85),which occurs naturally as nesquehonite,and a pentahydrate, MgCO3.5H2O (monoclinic; r.d. 1.73),which occurs as lansfordite. Magnesiumcarbonate also occurs in themixed salt dolomite (CaCO3.MgCO3)and as basic magnesium carbonatein the two minerals artinite(MgCO3.Mg(OH)2.3H2O) and hydromagnesite(3MgCO3.Mg(OH)2.3H2O).The anhydrous salt can be formed byheating magnesium oxide in astream of carbon dioxide:
MgO(s) + CO2(g) → MgCO3(s)
Above 350°C, the reverse reactionpredominates and the carbonate decomposes.Magnesium carbonate isused in making magnesium oxideand is a drying agent (e.g. in tablesalt). It is also used as a medicalantacid and laxative (the basic carbonateis used) and is a componentof certain inks and glasses.
MgO(s) + CO2(g) → MgCO3(s)
Above 350°C, the reverse reactionpredominates and the carbonate decomposes.Magnesium carbonate isused in making magnesium oxideand is a drying agent (e.g. in tablesalt). It is also used as a medicalantacid and laxative (the basic carbonateis used) and is a componentof certain inks and glasses.
Definition
hydromagnesite: A mineral formof basic magnesium carbonate,3MgCO3.Mg(OH)2.3H2O.
Definition
lansfordite: A mineral form ofmagnesium carbonate pentahydrate,MgCO3.5H2O.
Definition
The term magnesite is loosely used as a synonym for magnesia as are also the terms caustic-calcined magnesite, dead-burned magnesite, and synthetic magnesite.
General Description
White, yellowish, grayish-white or brown crystalline solid or crystalline powder. Density: 3-3.1 g cm-3. An important ore for magnesium. Used in the manufacture of materials capable of withstanding very high temperatures. Sometimes used to produce carbon dioxide.
Reactivity Profile
Magnesium carbonate has generally low chemical reactivity. Non-flammable and non-combustible. Reacts with acids and acidic salts to generate gaseous carbon dioxide with effervescence (bubbling). The reaction may be rapid and exothermic with concentrated solutions of acids. The efferversence can create foaming. Incompatible with formaldehyde.
Hazard
A nuisance particulate.
Agricultural Uses
Hydromagnesite is a magnesium ore which occurs as a carbonate. Magnesium carbonate occurs in a mixed salt dolomite (CaCO3.MgCO3) and as basic magnesium carbonate in two minerals, namely, artinite (MgCO3.Mg(OH)2.3H2O) and hydromagnesite (3MgCO3.Mg(OH)2.3H2O).
Agricultural Uses
Magnesium carbonate is a white compound occurring in anhydrous and hydrated forms. It is used as a fertilizer and also for making magnesium oxide.
The anhydrous material is found naturally in mineral magnesite. There is also a trihydrate, MgC03·3H2O (rhombic) that occurs naturally as nesquehonite, and a pentahydrate, MgCO3.5H2O (monoclinic) that occurs as lansfordite.
Magnesium carbonate also occurs in a mixed salt dolomite (CaCO3·MgCO3) and as basic magnesium carbonate in two minerals, namely, artinite [MgCO3·Mg(OH)2.3H2O] and hydromagnesite [3MgC03·Mg(OH)2.3H2O]. Heating magnesium oxide in a stream of carbon dioxide leads to the formation of the anhydrous salt.
Above 350°C, the reverse reaction predominates and the carbonate decomposes to give back MgO.
The anhydrous material is found naturally in mineral magnesite. There is also a trihydrate, MgC03·3H2O (rhombic) that occurs naturally as nesquehonite, and a pentahydrate, MgCO3.5H2O (monoclinic) that occurs as lansfordite.
Magnesium carbonate also occurs in a mixed salt dolomite (CaCO3·MgCO3) and as basic magnesium carbonate in two minerals, namely, artinite [MgCO3·Mg(OH)2.3H2O] and hydromagnesite [3MgC03·Mg(OH)2.3H2O]. Heating magnesium oxide in a stream of carbon dioxide leads to the formation of the anhydrous salt.
Above 350°C, the reverse reaction predominates and the carbonate decomposes to give back MgO.
Agricultural Uses
Nesquehonite is the natural form of magnesium carbonate trihydrate (MgCO3.3H2O).
Pharmaceutical Applications
As an excipient, magnesium carbonate is mainly used as a directly compressible tablet diluent in concentrations up to 45% w/w. Heavy magnesium carbonate produces tablets with high crushing strength, low friability, and good disintegration properties. However, magnesium carbonate can have varying effects on dissolution and stability.Magnesium carbonate has been incorporated in microsphere formulations for the purpose of stabilizing encapsulated proteins. It has also been coencapsulated in poly(lactide-co-glycolide) microsphere formulations to neutralize acidity and enhance the immunogenicity of a contraceptive peptide vaccine. Magnesium carbonate is also used to absorb liquids, such as flavors, in tableting processes. Magnesium carbonate is additionally used as a food additive and therapeutically as an antacid.
Safety
Magnesium carbonate is used as an excipient in oral solid-dosage pharmaceutical formulations and is generally regarded as an essentially nontoxic and nonirritant material. However, the use of magnesium salts, such as magnesium carbonate, is contraindicated in patients with renal impairment. In certain studies, magnesium carbonate has been shown to be an effective phosphate binder in short-term use for patients with chronic kidney disease, but the effects of long-term use require further study.The probable oral lethal dose in humans has been estimated at 0.5–5.0g/kg bodyweight. On contact with gastric acid, magnesium carbonate reacts in the stomach to form soluble magnesium chloride and carbon dioxide. Magnesium carbonate should therefore not be used as an antacid by those individuals whose stomachs cannot tolerate the evolution of carbon dioxide. Some magnesium is absorbed but is usually excreted in the urine. As with other magnesium salts, magnesium carbonate has a laxative effect and may cause diarrhea.
Therapeutically, the usual dose of magnesium carbonate as an antacid is 250–500mg, and 2.0–5.0g as a laxative.
Therapeutically, the usual dose of magnesium carbonate as an antacid is 250–500mg, and 2.0–5.0g as a laxative.
storage
Magnesium carbonate is stable in dry air and on exposure to light. The bulk material should be stored in a well-closed container in a cool, dry place.
Incompatibilities
Incompatible with phenobarbital sodium,diazepam solution at a pH≥5, some binary powder mixtures, lansoprazole, and formaldehyde. Acids will dissolve magnesium carbonate, with the liberation of carbon dioxide. Slight alkalinity is imparted to water. Magnesium carbonate was also found to increase the dissolution of acetazolamide formulations at a pH of 1.12; however, dissolution was retarded at a pH of 7.4.
Regulatory Status
GRAS listed. Accepted as a food additive in Europe. Included in the FDA Inactive Ingredients Database (oral capsules and tablets). Included in nonparenteral medicines licensed in the UK.
Company Profile Introduction
Established in 2014,Career Henan Chemical Co. is a manufacturerspecializing in the sale of fine chemicals.
Mainly deals in the sales of:
Pharmaceutical intermediates
OLED intermediates:
Pharmaceutical intermediates;
OLED intermediates;
Recommended supplier
-
VIP1年
- Sudeep Pharma Pvt Ltd
- 546-93-0 98%
- Inquiry
- 2024-03-21
-
VIP1年
- Madhav Snow White Industries
- 546-93-0 Magnesium carbonate 99%
- Inquiry
- 2024-03-12
-
VIP1年
- Ams Fine Chemicals
- 546-93-0 Magnesium carbonate 98%
- Inquiry
- 2024-02-20
-
VIP1年
- Melox Chemicals
- Magnesium carbonate 546-93-0 98%
- Inquiry
- 2024-02-15
-
VIP1年
- Par Drugs and Chemicals Ltd
- Magnesium carbonate 99%
- Inquiry
- 2024-02-14
-
VIP1年
- Prakash Chemicals Agencies
- 546-93-0 99%
- Inquiry
- 2024-02-14
-
VIP1年
- Elite Chemicals
- 546-93-0 Magnesium carbonate 98%
- Inquiry
- 2024-02-14
-
VIP1年
- Orrery Enterprise
- Magnesium carbonate 546-93-0 98%
- Inquiry
- 2024-02-09
-
VIP1年
- Hari Om Chemicals
- Magnesium carbonate 98%
- Inquiry
- 2024-02-08
-
VIP1年
- Nitika Pharmaceutical Specialities Pvt Ltd
- 546-93-0 98%
- Inquiry
- 2024-02-05
- Since:2014-12-17
- Address: Room 702, Floor 7, Building 10, National University Science Park, High-Tech Zone, Zhengzhou City, H
INQUIRY
