

Dapagliflozin
| Price | USD1.00 |
| Packge | 25KG |
- Min. Order:1KG
- Supply Ability:100KG
- Time:2019-07-06
Product Details
- Product NameDapagliflozin
- CAS No. 461432-26-8
- EINECS No.1592732-453-0
- MFC21H25ClO6
- MW408.88
- AppearanceSolidWhite to Pale Yellow
- density 1.349
- Boiling point 609.0±55.0 °C(Predicted)
- storage temp. 2-8°C
- Melting point 55-58°C
CR084
| Dapagliflozin Basic information |
| Product Name: | Dapagliflozin |
| Synonyms: | DAPAGLIFLOZIN;(1S)-1,5-Anhydro-1-C-[4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl]-D-glucitol;2-(3-(4-Ethoxybenzyl)-4-chlorophenyl)-6-hydroxymethyltetrahydro-2H-pyran-3,4,5-triol;Bms 512148;D-Glucitol, 1,5-anhydro-1-C-(4-chloro-3-((4-ethoxyphenyl)methyl)phenyl)-, (1S)-;Dapagliflozin(BMS-512148);(2S,4R,5R)-2-(4-chloro-3-(4-ethoxybenzyl)phenyl)-6-(hydroxyMethyl)tetrahydro-2H-pyran-3,4,5-triol;1-[4-Chloro-3-(4-ethoxybenzyl)phenyl]-1-deoxy-beta-D-glucopyranose (1S)-1,5-Anhydro-1-C-[4-chloro-3-(4-ethoxybenzyl)phenyl]-D-glucitol (2S,3R,4R,5S,6R)-2-[4-Chloro-3-(4-ethoxybenzyl)phenyl]-6-(hydroxyMethyl)tetrahydro-2H-pyran-3,4,5-triol |
| CAS: | 461432-26-8 |
| MF: | C21H25ClO6 |
| MW: | 408.875 |
| EINECS: | 1592732-453-0 |
| Product Categories: | APIs;Aromatics;Heterocycles;Intermediates & Fine Chemicals;Pharmaceuticals;Other APIs;Inhibitors |
| Mol File: | 461432-26-8.mol |
 |
|
| Dapagliflozin Chemical Properties |
| density | 1.349 |
| Safety Information |
| Dapagliflozin Usage And Synthesis |
| Diabetes drugs | Dapagliflozin (ForxigaTM) is a new antidiabetic drug jointly developed by Bristol-Myers Squibb and AstraZeneca, being approved by the European Medicines Agency (EMA) on November 12, 2012. It is also the first approved SGLT2 inhibitor for the treatment of type II diabetes, being an important option in the treatment of diabetes, and is used to improve glycemic control as an adjunct to dietary and exercise for adults with type II diabetes. Dapagliflozin is a sodium-glucose co-transporter 2 inhibitor. On January 8, 2014, the US Food and Drug Administration (FDA) have approved it for being used in the treatment of type II diabetes. Meanwhile, FDA requires the producers to conduct post-marketing research on drug-related risks. The post-marketing trial requested by the FDA includes a cardiovascular outcome trial for assessing the cardiovascular risk for high-risk patients after treatment with dapagliflozin at baseline and a study to assess the risk of bladder cancer in recruited patients. Another study will assess the bladder tumor-promoting effect of this drug on rodent animals. Two studies will assess the pharmacokinetics, efficacy and safety of dapagliflozin in pediatric patients; a set of strengthened pharmacovigilance program will monitor liver abnormalities and pregnancy outcome reports in patients receiving daglitazone. Dapagliflozin will be marketed under the tradename Farxiga by Haoeyou Pharmacy. The above information is edited by Andy of chemicalbook. |
| Pharmacological effects | Dapagliflozin works through inhibiting sodium-glucose transporter 2 (SGLT2), a protein in the kidney that reabsorbs glucose into the bloodstream. This allows extra glucose to be excreted through the urine, improving glycemic control without increasing insulin secretion. The use of this drug requires patients with normal renal function while patients of moderate to severe renal insufficiency should be disabled to use this drug. Single application of this product or combination with metformin, pioglitazone, glimepiride, insulin and other drugs can significantly reduce the HbA1c and fasting blood glucose of patients suffering type II diabetes. The frequency of the adverse reaction was similar to placebo with low risk of hypoglycemia, being able to reduce body weight. The efficacy of dapagliflozin is comparable with the dipeptidyl peptidase inhibitors, and several new hypoglycemic drugs, and can also mildly lower the blood pressure and body weight. The drug has 5mg and 10mg two tablets to choose from, can be either used alone or together with insulin, including other diabetes drugs. |
| Pharmacokinetics | In healthy subjects, dapagliflozin was rapidly absorbed after oral administration with a peak time Tmax being 1 to 2 hours, a protein binding rate of 91%, an oral bioavailability of about 78% and a plasma terminal half-life of 12.9 hours. After oral administration, the drug is mainly metabolized by the uridine diphosphate glucuronosyltransferase 1A9 (UGT1A9) into the inactive metabolite in the liver with the smaller part being metabolized by the P450 enzyme and of no inhibitory or inducing effect on the P450 enzyme. Drug prototypes and related metabolites were excreted through urine (75%) and faeces (21%). Compare simultaneous administration of this product with high-fat food and with the fasting administration, Tmax can be extended by 1-fold, but the absorption did not affect the degree, so can be administrated together with the food. The pharmacokinetics of daglitazone was significantly affected by renal function. Diabetic patients with mild, moderate or severe renal insufficiency are merged to be subject to oral administration of 20 mg • d-1 daglitazone for 7 days. The mean systemic exposure amount, compared with patients with normal renal function, is respectively 32%, 60% and 87% higher. For patients with normal renal function, mild insufficiency, moderate insufficiency and severe insufficiency, the urinary glucose excretion amount in 24 hours of steady state was 85, 52, 18 and 11g, successively. Kasichayanula et al have studied the pharmacokinetic effects of liver dysfunction on daglitazone. The patients with mild, moderate and severe hepatic insufficiency having a single oral dose of 10 mg of daglitazone, the Cmax of each group was 12% lower, 12% higher and 40% higher than that with normal liver function, respectively. The AUC of each group was significantly higher than that of normal liver function by 3%, 36% and 67%. Therefore, it is not recommended to apply daglitazone to patients of moderate and severe renal dysfunction. Severe liver dysfunction patients need to reduce the use of dose. |
| Synthesis method | 5-bromo-2-chlorobenzoic acid is subject to acylating chlorination, and has Friedel-Crafts reaction with phenylethyl ether for reduction of its carbonyl group, generating 5-bromo-2-chloro-4'-ethoxydiphenyl methane, further subjecting to condensation with 2, 3, 4, 6-tetra-O-trimethylsilyl-D-glucopyranosanoic acid-1,5-lactone. The anomeric carbon hydroxyl group is subject to etherification and deprotection to give 2-chloro-5-(1-methoxy-D-glucopyranose-I-yl)-4'-ethoxydiphenylmethane, and then use Et3SiH/BF3 • OEt2 for reduction to remove methoxy, followed by acetic anhydride esterification and hydrolysis to give hypoglycemic agents daglitazone with the overall yield of about 40%.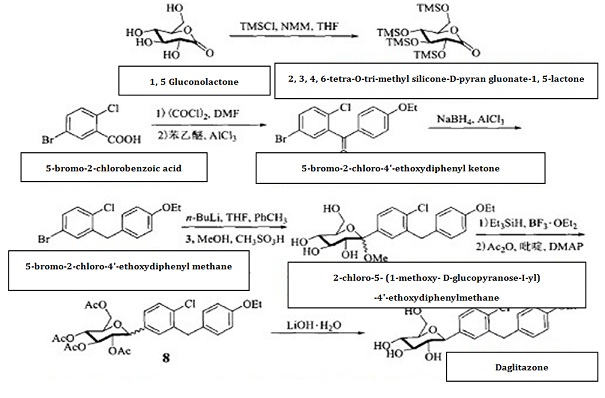 Fig.1 shows the chemical reaction route of synthesizing dapagliflozin. |
| Safety | Daglitazone has excellent tolerance and safety with the incidence of adverse events associated with 10 mg • d-1 daglitazone being similar to that of placebo. Common adverse events included hypoglycemia, polyuria, back pain, genital infections, urinary tract infections, dyslipidemia and hematocrit (HCT) increase. The overall risk of hypoglycemia is low, and the incidence of hypoglycemia is associated with other basic hypoglycemic agents. The incidence of hypoglycemia was higher in patients subjecting to joint treatment between daglitazone and sulfonylureas or insulin compared with placebo. Therefore, when this product is used in combination with insulin or insulin secretagogue, you may need to adjust the dose of the latter one. |
| Drug interactions | This product is mainly metabolized in the liver by UGT1A9 metabolism, being the P-glycoprotein substrate. Study confirmed that the pharmacokinetics of daglitazone was not affected by metformin, pioglitazone, sitagliptin, glimepiride, voglibose, and simvastatin, valsartan, warfarin, and digoxin. The serum concentrations of the above-mentioned drugs are also not clinically significantly affected by daglitazone. Rifampicin can reduce the exposure amount of daglitazone by 22% while mefenamic acid can increase the body exposure amount by 51%, but have no clinically significant effect on 24 h urine glucose excretion. |
| Chemical Properties | White Solid |
| Uses | therapeutic for diabetes I or II, and hyperglycemia |
| Uses | A sodium-glucose transporter 2 inhibitor. |
| Definition | ChEBI: A C-glycosyl comprising beta-D-glucose in which the anomeric hydroxy group is replaced by a 4-chloro-3-(4-ethoxybenzyl)phenyl group. Used (in the formo f its propanediol monohydrate) to improve glycemic ontrol, along with diet and exercise, in adults with type 2 diabetes. |
Company Profile Introduction
Henan CoreyChem Co., Ltd, based on the original Zhengzhou Cote Chemical Research Institute, be brave in absorbing highly educated talents & overseas returnees; actively responded to Zhengzhou City High-tech Zone Government’s Special Care Policy, reorganized and founded in National University of Science and Technology Park, which is a high-tech, stock enterprise of high-end chemical Custom synthesis;The park was created by the People's Government of Henan Province, and proved by Ministry of Education and the National Science & Technology, taking the construction mode of "many college a park, and common development", mainly depends on Zhengzhou University and Henan University’s scientific research and talent advantage to set up Universities, scientific research institute and enterprise scientific research achievements transformation platform, to make high-tech enterprises incubate, is the new high-tech talent gathering base, high and new technology industry enterprise radiation base, colleges and universities technological innovation base.
Henan Coreychem Co., Ltd, facing global High-tech pharmaceutical raw materials, high complex new type intermediates, fine chemicals custom synthesis, scale-up production and Rare chemicals trade. Corey have well-equipped machine, strong technical force and considerate marketing team service. We also have rich experience advantage in basic research, small scale process development, scale-up, industrial technology development & production and cost control.
Recommended supplier
-
VIP1年
- AKASH PHARMA EXPORTS
- Dapagliflozin 99%
- Inquiry
- 2024-05-16
-
VIP1年
- Molsyns Research
- Dapagliflozin Reference Standard 98%
- Inquiry
- 2024-04-01
-
VIP1年
- Raks Pharma Pvt Ltd
- 461432-26-8 Dapagliflozin 99%
- Inquiry
- 2024-03-27
-
VIP1年
- Aspen Biopharma Labs Pvt Ltd
- Dapagliflozin 98%
- Inquiry
- 2024-03-16
-
VIP1年
- Sibram Pharmaceutical
- 461432-26-8 99%
- Inquiry
- 2024-03-16
-
VIP1年
- Turtle Pharma Private Limited
- 461432-26-8 98%
- Inquiry
- 2024-03-15
-
VIP1年
- Vijaya Pharma And Life Science
- Dapagliflozin 98%
- Inquiry
- 2024-03-13
-
VIP1年
- Rini Life Science Pvt Ltd
- 461432-26-8 99%
- Inquiry
- 2024-03-02
-
VIP1年
- Bioaltus Laboratories Pvt Ltd
- 461432-26-8 Dapagliflozin 98%
- Inquiry
- 2024-03-01
-
VIP1年
- Suvan LifeSciences (formerly Sansh Biotech Pvt Ltd)
- Dapagliflozin 461432-26-8 98%
- Inquiry
- 2024-02-29
- Since:2014-12-17
- Address: No.967,15th Floor,Unit 7, Building 1, No.70 of DianChang Road, High-tech Development Zone, Zhengzho
INQUIRY
