PIROMIDIC ACID
- CAS NO.:19562-30-2
- Empirical Formula: C14H16N4O3
- Molecular Weight: 288.3
- MDL number: MFCD00058574
- EINECS: 243-161-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-11-19 23:02:33
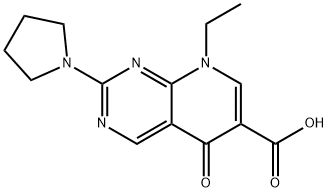
What is PIROMIDIC ACID?
Description
Piromidic acid is a quinolone antibiotic that is active against Gram-positive and Gram-negative bacteria including S. aureus and E. coli (MICs = 10 and 1 μg/ml, respectively). It inhibits growth of nalidixic acid-sensitive strains of E. coli in an in vitro model of bacterial cystitis when used at concentrations of 10 and 50 mg/L. Piromidic acid also has antimalarial properties and is active against chloroquine-sensitive and -resistant strains of P. falciparum in vitro (IC50s = 41.4 and 14.4 μg/ml, respectively) as well as against hepatic stages of P. yoelii yoelii (IC50 = 21.6 μg/ml).
Originator
Panacid, Dainippon, Japan ,1972
The Uses of PIROMIDIC ACID
Piromidic acid is a quinolone antibacterial.
The Uses of PIROMIDIC ACID
Antibacterial;Topoisomerase II inhibitor
What are the applications of Application
Piromidic Acid is gram-negative antimicrobial
Definition
ChEBI: Piromidic acid is a pyridopyrimidine that is 5-oxo-5,8-dihydropyrido[2,3-d]pyrimidine-6-carboxylic acid, substituted at position 2 by a pyrrolidin-1-yl group and at position 8 by an ethyl group. A synthetic antibacterial which is used for the treatment of urinary tract and intestinal infections. It has a role as an antibacterial drug and a DNA synthesis inhibitor. It is a quinolone antibiotic, a member of pyrrolidines, a tertiary amino compound, a monocarboxylic acid and a pyridopyrimidine.
Manufacturing Process
150 mg of 6-carboxy-5,8-dihydro-8-ethyl-2-methylthio-5-oxopyrido[2,3d]pyrimidine was added to 30 ml of absolute ethanol containing 1.1 g of dissolved pyrrolidine, and the mixture was reacted for 5 hours at 95°C in a sealed tube. The solvent was removed by distillation, and the residue was recrystallized from methanol-chloroform. There were obtained 111 mg of 6carboxy-5,8-dihydro-8-ethyl-5-oxo-2-pyrrolidino-pyrido[2,3-d]pyrimidine having a MP of 314° to 316°C.
The starting material is produced by reacting 6-amino-2-methylthiopyrimidine with ethoxymethylene malonic acid diethyl ester. That intermediate is thermally treated in diphenyl ether to give 6-ethoxycarbonyl-2-methylthio-5oxo-5,8-dihydro-pyrido[2,3-d]pyrimidine. The ethoxy group is hydrolyzed off with sodium hydroxide and one nitrogen is ethylated with diethyl sulfate to give the starting material. These are the same initial steps as used in the pipemidic acid syntheses earlier in this volume.
Therapeutic Function
Antibacterial (urinary)
Pharmaceutical Applications
A pyrimidopyrimidine derivative with a C7-pyrrolidinyl ring, allowing a slight increase in activities against Gram-positive cocci. Its main antibacterial activity is close to that of nalidixic acid and there have been reports of renal toxicity. It is available in only a few countries.
Safety Profile
Poison by subcutaneous and intravenous routes. Moderately toxic by skin contact and intraperitoneal routes. An antibacterial agent. When heated to decomposition it emits toxic fumes of NOx.
Properties of PIROMIDIC ACID
| Melting point: | 314-316° |
| Boiling point: | 430.54°C (rough estimate) |
| Density | 1.1895 (rough estimate) |
| refractive index | 1.6000 (estimate) |
| storage temp. | 0-6°C |
| solubility | Chloroform (Slightly, Heated, Sonicated), Ethanol (Slightly, Heated), Methanol ( |
| form | Solid |
| pka | 4.14±0.20(Predicted) |
| color | Crystals from EtOH/CHCl3 |
Safety information for PIROMIDIC ACID
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for PIROMIDIC ACID
New Products
Tert-butyl bis(2-chloroethyl)carbamate 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid 1,1’-CARBONYLDIIMIDAZOLE DIETHYL AMINOMALONATE HYDROCHLORIDE R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) N-METHYL INDAZOLE-3-CARBOXYLIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran
You may like
-
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1 -
 733039-20-8 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 98+View Details
733039-20-8 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 98+View Details
733039-20-8