Org 31540
- CAS NO.:104993-28-4
- Empirical Formula: C31H53N3O49S8
- Molecular Weight: 1508.26
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-10-23 13:36:13
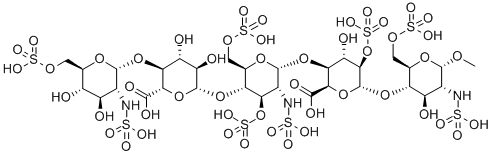
What is Org 31540?
Absorption
100% bioavailability when administered subcutaneously
Toxicity
As with other anticoagulants, the main concern is increased bleed risk. The risk of hemorrhage may increase with decreased renal function, body mass less than 50 kg, and moderate to severe hepatic function.
Background
Fondaparinux (Arixtra) is a synthetic anticoagulant agent consisting of five monomeric sugar units and a O-methyl group at the reducing end of the molecule. It is structurally similar to polymeric glycosaminoglycan heparin and heparan sulfate (HS) when they are cleaved into monomeric units. The monomeric sequence in heparin and HS is thought to form the high affinity binding site for the natural anti-coagulant factor, antithrombin III (ATIII). Once bound to heparin or HS, the anticoagulant activity of ATIII is potentiated by 1000-fold. Fondaparinux potentiates the neutralizing action of ATIII on activated Factor X 300-fold. Fondaparinux may be used: to prevent venous thromboembolism in patients who have undergone orthopedic surgery of the lower limbs (e.g. hip fracture, hip replacement and knee surgery); to prevent VTE in patients undergoing abdominal surgery who are are at high risk of thromboembolic complications; in the treatment of deep vein thrombosis (DVT) and pumonary embolism (PE); in the management of unstable angina (UA) and non-ST segment elevation myocardial infarction (NSTEMI); and in the management of ST segment elevation myocardial infarction (STEMI).
Indications
Approved for: (1) prophylaxis of VTE for up to one month post surgery in patients undergoing orthopedic surgery of the lower limbs such as hip fracture, hip replacement and knee surgery; (2) prophylaxis of VTE patients undergoing abdominal surgery who are at high risk of thromboembolic complications (e.g. patients undergoing abdominal cancer surgery); (3) treatment of acute DVT and PE; (4) management of UA and NSTEMI for the prevention of death and subsequent myocardial infarction (MI); and (5) management of STEMI for the prevention of death and myocardial reinfarction in patients who are managed with thrombolytics or who are initially to receive no form of reperfusion therapy. Fondaparinux should not be used as the sole anticoagulant during percutaneous coronary intervention (PCI) due to an increased risk of guiding catheter thrombosis.
Definition
ChEBI: A synthetic pentasaccharide which, apart from the O-methyl group at the reducing end of the molecule, consists of monomeric sugar units which are identical to a sequence of five monomeric sugar units that can be isolated after either che ical or enzymatic cleavage of the polymeric glycosaminoglycans heparin and heparan sulfate.
Pharmacokinetics
Fondaparinux binds specifically to the natural anticoagulant factor, ATIII. Binding to ATIII potentiates the neutralizing action of ATIII on Factor Xa 300-times. Neutralization of Factor Xa decreases the conversion of prothrombin to thrombin, which subsequently decreases the conversion of fibrinogen to fibrin (loose meshwork). The decrease in thrombin also decreases the activation of Factor XIII, which decreases the conversion of fibrin in its loose meshwork form to its stabilized meshwork form. Disruption of the coagulation cascade effectively decreases the formation of blood clots. Fondaparinux does not inactivate thrombin (activated Factor II). According to the manufacturer, fondaparinux has no known effect on platelet function. In studies comparing fondaparinux to enoxaparin, decreases in platelet levels were observed in similar numbers of patients from both groups (2-5%) . At the recommended dose, Fondaparinux does not affect fibrinolytic activity or bleeding time. There is no antidote for fondaparinux. Monitoring of the anticoagulant activity of fondaparinux is not generally required. The anti-factor Xa assay may be used to monitor therapy in special populations such as those with renal impairment or who are pregnant. Complete blood count (CBC) and kidney function should be monitored during treatment.
Metabolism
Not metabolized
Properties of Org 31540
| Density | 2.27±0.1 g/cm3(Predicted) |
| pka | -3?+-.0.18(Predicted) |
Safety information for Org 31540
Computed Descriptors for Org 31540
Org 31540 manufacturer
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5,6-Dimethoxyindanone 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateYou may like
-
 104993-28-4 Fondaparinux 98%View Details
104993-28-4 Fondaparinux 98%View Details
104993-28-4 -
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1