N4-Acetylcytidine
- CAS NO.:3768-18-1
- Empirical Formula: C11H15N3O6
- Molecular Weight: 285.25
- MDL number: MFCD00006540
- EINECS: 223-195-6
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-26 12:07:08
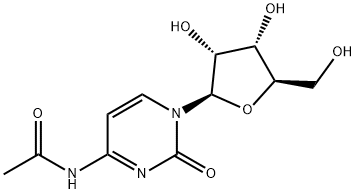
What is N4-Acetylcytidine?
Description
N4-Acetylcytidine is a modified nucleoside and endogenous urinary nucleoside product of the degradation of tRNA. N4-Acetylcytidine is a biological marker for various cancers with elevated concentrations present in urine. tRNA has been shown to be excreted in abnormal amounts in the urine of cancer patients. tRNA from neoplastic tissue had a much more rapid turnover rate than the tRNA from the corresponding normal tissue. Evidence indicates that methylation of tRNA occurs only after synthesis of the intact macromolecule. Because there are no specific enzyme systems to incorporate the modified nucleosides into the macromolecular nucleic acid, these nucleosides once released in the process of tRNA turnover cannot be reutilized, nor are they further degraded, but are excreted in urine.
N4-Acetylcytidine is also a partially protected cytidine and therefore can be used as a synthetic building block to prepare further derivatized nucleosides such as 2’,3’-dideoxycytidine.
The Uses of N4-Acetylcytidine
N4-Acetylcytidine (cas# 3768-18-1) is a nucleotide-derived metabolite used as biomarkers for diagnosis of inflammation-related diseases.
Definition
ChEBI: N(4)-acetylcytidine is cytidine in which one of the exocyclic amino hydrogens is substituted by an acetyl group. It has a role as a metabolite. It is a member of cytidines, a member of acetamides and a secondary carboxamide.
Biosynthesis
N4-acetylcytidine (ac4C) is a post-transcriptional modification of RNA that is conserved across all domains of life. All characterized sites of ac4C in eukaryotic RNA occur in the central nucleotide of a 5’-CCG-3’ consensus sequence. However, the thermodynamic consequences of cytidine acetylation in this context have never been assessed due to its challenging synthesis. Here we report the synthesis and biophysical characterization of ac4C in its endogenous eukaryotic sequence context. First, we develop a synthetic route to homogenous RNAs containing electrophilic acetyl groups. Next, we use thermal denaturation to interrogate the effects of ac4C on duplex stability and mismatch discrimination in a native sequence found in human ribosomal RNA. Finally, we demonstrate the ability of this chemistry to incorporate ac4C into the complex modification landscape of human tRNA, and use duplex melting combined with sequence analysis to highlight a potentially unique enforcing role for ac4C in this setting. By enabling the analysis of nucleic acid acetylation in its physiological sequence context, these studies establish a chemical foundation for understanding the function of a universally-conserved nucleobase in biology and disease.
Site-Specific Synthesis of N4-Acetylcytidine in RNA Reveals Physiological Duplex Stabilization
https://www.biorxiv.org/content/10.1101/2021.11.12.468326v1.full.pdf
Properties of N4-Acetylcytidine
| Melting point: | 199 °C (dec.)(lit.) |
| Density | 1.72±0.1 g/cm3(Predicted) |
| storage temp. | Sealed in dry,Store in freezer, under -20°C |
| solubility | DMSO (Slightly, Heated), Methanol (Slightly, Heated) |
| form | Solid |
| pka | 10.21±0.20(Predicted) |
| color | White to Pale Yellow |
| CAS DataBase Reference | 3768-18-1(CAS DataBase Reference) |
| NIST Chemistry Reference | Cytidine, n-acetyl-(3768-18-1) |
Safety information for N4-Acetylcytidine
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P280:Wear protective gloves/protective clothing/eye protection/face protection. |
Computed Descriptors for N4-Acetylcytidine
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran



You may like
-
 N4-Acetylcytidine 95% CAS 3768-18-1View Details
N4-Acetylcytidine 95% CAS 3768-18-1View Details
3768-18-1 -
 3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details
3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details -
 614-19-7 98%View Details
614-19-7 98%View Details
614-19-7 -
 3112-85-4 Methyl phenyl sulfone 98%View Details
3112-85-4 Methyl phenyl sulfone 98%View Details
3112-85-4 -
 20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 -
 3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details
3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details -
 57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 -
 4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
5305-40-8
