Iodic acid
- CAS NO.:7782-68-5
- Empirical Formula: HIO3
- Molecular Weight: 175.91
- MDL number: MFCD00011348
- EINECS: 231-962-1
- Update Date: 2025-01-27 09:38:02
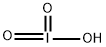
What is Iodic acid?
Chemical properties
colorless, rhomb crystal(s) or white, crystal(s) powder(s); darkens on exposure to light; it is a moderately strong acid; used in analytical chemistry and in medicine [HAW93] [MER06]
Physical properties
Iodic acid, HIO3, can be obtained as a white solid. It
dissolves in water very well, but it also exists in the
pure state, as opposed to chloric acid or bromic acid.
Iodic acid contains iodine in the oxidation state+5
and it is one of the most stable oxo-acids of the halogens
in its pure state. When iodic acid is carefully heated, it
dehydrates to iodine pentoxide. On subsequent heating,
the iodine pentoxide further decomposes, giving
a mixture of iodine, oxygen and lower oxides of iodine.
Iodic acid can be produced by oxidizing I2 with
chlorine in an aqueous solution. Iodic acid can be
used to synthesize sodium or potassium iodate salts
(which are used in salt as a source of iodine in the
human body.
The Uses of Iodic acid
Iodic acid is used in analytical chemistry laboratories to standardize solutions of both weak and strong bases using methyl red or methyl orange as the indicator. It acts as a key starting material to synthesize sodium or potassium iodate, thereby increasing the iodine content of salt.
The Uses of Iodic acid
A strong acid in analytical chemistry.
What are the applications of Application
Iodic acid is a strong acid in analytical chemistry
Definition
iodic acid: Any of various oxoacids of iodine, such as iodic(V) acid and iodic(VII) acid. When used without an oxidation state specifled, the term usually refers to iodic(V) acid (HIO3).
Preparation
Iodic acid may be prepared by the reaction of sulfuric acid with bariumiodate. The solution is filtered to remove barium sulfate and then crystallizedto obtain iodic acid:
Ba(IO3)2 + H2SO4 → BaSO4 + 2HIO3
It also may be produced by oxidation of iodine with concentrated nitric acid:
3I2 + 10HNO3 → 6HIO3 + 10NO + 2H2O
Also, iodic acid may be obtained by oxidation of iodine with chlorine in diluteacidic solutions:
I2 + 5Cl2 + 6H2O → 2HIO3 + 10HCl
Another method of preparation involves oxidation of iodine with hydrogenperoxide:
I2 + 5H2O2 → 2HIO3 + 4H2O
It also may be prepared by treating hypoiodous acid with a base:
3HIO + 2OH¯ → HIO3 + 2H2O + I¯
Hypoiodous acid may be obtained by alkaline hydrolysis of iodine at pH 12:
I2 + H2O → HIO + H+ + I¯
Iodic acid dehydrates to iodine pentaoxide when heated at 180°C:
2HIO3 → I2H5 + H2O
Iodic acid is a relatively weak monoprotic acid, the Ka value at 25°C is 1.6x10-1. Several species have been detected in concentrated aqueous solutions,which include IO3-, H+, HIO3, (HIO3)2 and (HIO3)3. Its solution turns blue lit-mus red and then bleaches the litmus paper because of its strong oxidizingproperties.
When heated with potassium iodate, potassium hydrogen iodate is formed:
HIO3 + KIO3 → KH(IO3)2
An aqueous solution of iodic acid is a strong oxidizing agent. It liberates iodine from iodides:
IO3¯ + 5I¯ + 6H+ → 3I2 + 3H2O or, HIO3 + 5HI → 3I2 + 3H2O
In an aqueous solution, iodic acid oxidizes hydrogen sulfide to sulfur:
2HIO3 + 5H2S → I2 + 6H2O + 5S
The solid iodic acid reacts vigorously with sulfur, phosphorus and other non-metals.
General Description
Iodic acid is the hydrated form of I2O5. Reaction of iodic acid with hydrogen iodide has been described by electrolytic dissociation theory. Combustion of mixture of chromic, iodic, sulfuric and phosphoric acids has been proposed. Its Raman spectra have been recorded. Vibrational assignment of IO3- has been evaluated.
Hazard
Toxic by ingestion, strong irritant to eyes and skin.
Purification Methods
Dissolve iodic acid in the minimum volume of hot dilute HNO3, filter and evaporate in a vacuum desiccator until crystals are formed. Collect the crystals and wash them with a little cold H2O and dry them in air in the dark. It is soluble in H2O: 269g/100mL at 20o and 295g/100mL at 40o. It is soluble in dilute EtOH and darkens on exposure to light. It is converted to HIO3.I2O5 on heating at 70o, but at 220o complete conversion to HIO3 occurs. [Lamb et al. J Am Chem Soc 42 1636 1920, Bray & Caulkins J Am Chem Soc 53 44 1931.]
Properties of Iodic acid
| Melting point: | 110°C (dec.) |
| Density | 4.63 g/cm3 |
| solubility | Water (Slightly) |
| pka | 1.28±0.53(Predicted) |
| form | Solid |
| color | White to off-white |
| Specific Gravity | 4.629 |
| Water Solubility | 269 g/100 mL (20 ºC) |
| Sensitive | Light Sensitive |
| Merck | 14,5012 |
| Stability: | Stable. Incompatible with strong reducing agents, alcohols, organic materials. Light-sensitive. |
| CAS DataBase Reference | 7782-68-5(CAS DataBase Reference) |
| EPA Substance Registry System | Iodic acid (7782-68-5) |
Safety information for Iodic acid
| Signal word | Danger |
| Pictogram(s) |
 Flame Over Circle Oxidizers GHS03  Corrosion Corrosives GHS05 |
| GHS Hazard Statements |
H272:Oxidising liquids;Oxidising solids H314:Skin corrosion/irritation |
| Precautionary Statement Codes |
P220:Keep/Store away from clothing/…/combustible materials. P280:Wear protective gloves/protective clothing/eye protection/face protection. P310:Immediately call a POISON CENTER or doctor/physician. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Iodic acid
| InChIKey | ICIWUVCWSCSTAQ-UHFFFAOYSA-N |
Iodic acid manufacturer
DNS Fine Chemicals and Laboratories (P) Ltd
Svaks Biotech India Pvt Ltd
New Products
Cycloleucine 1-Amino-1-cyclohexanecarboxylic acid 6-Bromo-3-iodo-1-methyl-1H-indazole 3-(2,4-Dimethoxybenzyl)dihydropyrimidine-2,4(1H,3H)-dione 7-Bromo-1H-indazole ELECTROLYTIC IRON POWDER 1-(2-Ethoxyethyl)-2-(piperidin-4-yl)-1H-benzo[d]imidazole hydrochloride Decanonitrile tert-butyl 4-(1H-benzo[d]iMidazol-2-yl)piperidine-1-carboxylate N,N'-diallyl-1,3-diaminopropanedihydrochloride 4-Ethylbenzylamine N-(5-Amino-2-methylphenyl)acetamide 4-bromo-3,5-dimethylbenzenesulfonyl chloride 5-Bromo-2,3-dimethoxypyridine 2-Chloro-3-nitropyridine 2-methoxy-4-methyl-5-nitro pyridine 2-iodo-5-bromo pyridine methyl 6-chloro-2-(chloromethyl)nicotinate 2-amino-4-methyl-5-nitro pyridine 5-Fluoro-2-Oxindole (9H-fluoren-9-yl)methyl carbonochloridate 2-methyl-5-nitroaniline Diethyl phosphite 8-hydroxy-2,2,14,14-tetramethylpentadecanedioic acidRelated products of tetrahydrofuran



You may like
-
 7782-68-5 Iodic acid 99%View Details
7782-68-5 Iodic acid 99%View Details
7782-68-5 -
 7782-68-5 98%View Details
7782-68-5 98%View Details
7782-68-5 -
 Iodic Acid CAS 7782-68-5View Details
Iodic Acid CAS 7782-68-5View Details
7782-68-5 -
 Iodic acid 99.5% AR CAS 7782-68-5View Details
Iodic acid 99.5% AR CAS 7782-68-5View Details
7782-68-5 -
 Iodic acid, GR 99% CAS 7782-68-5View Details
Iodic acid, GR 99% CAS 7782-68-5View Details
7782-68-5 -
 Iodic acid 7782-68-5 98%View Details
Iodic acid 7782-68-5 98%View Details
7782-68-5 -
 Iodic acid CAS 7782-68-5View Details
Iodic acid CAS 7782-68-5View Details
7782-68-5 -
 Iodic acid CAS 7782-68-5View Details
Iodic acid CAS 7782-68-5View Details
7782-68-5