HYDROGEN SELENIDE
- CAS NO.:7783-07-5
- Empirical Formula: H2Se
- Molecular Weight: 80.98
- MDL number: MFCD00011224
- EINECS: 231-978-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:15:30
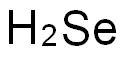
What is HYDROGEN SELENIDE?
Description
Hydrogen selenide is a colorless, flammablegas with a very offensive odor, resembling decayed horse-radish. The odor threshold is 0.3ppm (higher than the OCcu-pational exposure limits); many people rapidly lose theability to detect the odor of hydrogen selenide, so it is not areliable warning of exposure. Molecular weight = 80.98;Specific gravity (H2O:1)=2.1 (iquid); Boiling point =-41℃; FreezingMelting point= - 66℃; Relative vapordensity (air= 1): 2.80; Vapor pressure=9.5 atm at 21℃.Hazard Identification (based onNFPA-704 M RatingSystem): Health 4, Flammability 3, Reactivity 0. Soluble inwater; solubility= 0.9% at 22.7℃.
Chemical properties
colourless gas with a very unpleasant smell
Physical properties
Colorless gas; disagreeable odor; flammable; gas density 3.553 g/L; liquefiesat -41.25°C; liquid density 2.12 g/mL at -42°C; freezes at -65.73°C; criticaltemperature 137.85°C; critical pressure 88.0 atm; soluble in water, carbondisulfide and phosgene; pKa3.89 and 11.0 at 25°C, respectively, for the firstand second replaceable hydrogen.
The Uses of HYDROGEN SELENIDE
Preparation of metallic selenides and organoselenium compounds; in doping as mix for preparation of semiconductor materials containing controlled amounts of significant impurities.
Production Methods
Hydrogen selenide (H2Se) may be formed by the reaction of acids or water with metal selenides or wherever nascent hydrogen is in contact with soluble selenide compounds. It has no commercial use.
Preparation
Hydrogen selenide may be synthesized directly from elements by heatingat elevated temperatures. It is prepared by heating hydrogen at 440°C witheither selenium metal in a sealed tube or with selenium vapor over pumicestone:
H2 + Se → H2Se
Alternatively, the compound may be prepared by treating an alkali metalselenide with hydrochloric acid during mild heating:
NaSe + 2HCl → H2Se + NaCl
Definition
ChEBI: Selane is a selenium hydride and a mononuclear parent hydride. It has a role as an Escherichia coli metabolite and a mouse metabolite. It is a conjugate base of a selenonium. It is a conjugate acid of a selanide.
Air & Water Reactions
Highly flammable. Soluble in water.
Reactivity Profile
HYDROGEN SELENIDE is a colorless, flammable gas of unpleasant odor, a deadly poison by inhalation. HYDROGEN SELENIDE forms explosive mixtures with air. HYDROGEN SELENIDE reacts vigorously with strong oxidizing reagents, e.g., hydrogen peroxide, nitric acid [Sax, 9th ed., 1996, p.1843].
Hazard
Dangerous fire and explosion risk; reacts violently with oxidizing materials. Toxic by inhalation, strong irritant to skin, damaging to lungs and liver.
Health Hazard
HYDROGEN SELENIDE is a hazardous compound which can cause damage to the lungs and liver. Those experiencing dermatitis, chronic bronchitis, or any form of skin allergy or respiratory tract infection are at a greater risk.
Fire Hazard
HYDROGEN SELENIDE is extremely flammable; may be ignited by heat, sparks, or flames. Vapors may travel to a source of ignition and flash back. Container may explode in heat of fire. HYDROGEN SELENIDE is dangerous and forms explosive mixtures with air. HYDROGEN SELENIDE can decompose into toxic fumes. Incompatible with acid, water, halogenated hydrocarbons, oxidizers, hydrogen peroxide, and nitric acid. HYDROGEN SELENIDE should be stored out of the direct rays of the sun. Keep away from heat and flames.
Potential Exposure
Hydrogen selenide is used in semicon-ductor manufacture. Also, it may be produced by the reac-tion of acids or water and metal selenides, Or hydrogen and .soluble selenium compounds.
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek med-ical attention immediately. If this chemical contacts theskin, remove contaminated clothingand wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using uni versal precautions,including resuscitation mask) if breathing has stopped andCPR if heart action has stopped. Transfer promptly to amedical facility. When this chemical has been swallowed,get medical attention. Give large quantities of water andinduce vomiting. Medical observation is recommended for24- -48 h after breathing overexposure, as pulmonary edemamay be delayed. As first aid for pulmonary edema, a doctoror authorized paramedic may consider administering a corti-costeroid spray. Do not make an unconscious person vomit.If frostbite has occurred, seek medical attention immedi-ately; do NOT rub the affected areas or flush them withwater. In order to prevent further tissue damage, do NOTattempt to remove frozen clothing from frostbitten areas. Iffrostbite has NOT occured, immediately and thoroughlywash contaminated skin with soap and water.
storage
(2) Color Code- -Red Stripe:FlammabilityHazard: Store separately from all other flammable materi-als. (2) Color Code- Blue: Health Hazard/Poison: Store ina secure poison location. Prior to working with hydrogenselenide you should be trained on its proper handling andstorage. Before entering confined space where this chemical may be present, check to make sure that an explosive con-centration does not exist. Store at room temperature awayfrom contact with oxidizers, acids, water, and halogenatedhydrocarbons. Metal containers involving the transfer ofthis chemical should be grounded and bonded. Where possi-ble, automatically pump liquid from drums or other storagecontainers to process containers. Drums must be equippedwith self-closing valves, pressure vacuum bungs, and flamearresters. Use only nonsparking tools and equipment, espe-cially when opening and closing containers of this chemi-cal. Sources of ignition, such as smoking and open flames,are prohibited where this chemical is used, handled, orstored in a manner that could create a potential fire orexplosion hazard. Wherever this chemical is used, handled,manufactured, or stored, use explosion-proof electricalequipment and fittings. Procedures for the handling, use,and storage of cylinders should be in compliance withOSHA 1910.101 and 1910.169, as with the recommenda-tions of the Compressed Gas Association.
Shipping
Hydrogen selenide requires a shipping label of“POISON GAS, FI AMMABLE GAS.”It falls in HazardClass 2.3. It is a violation of transportation regulations torefill compressed gas cylinders without the express writtenpermission of the owner.
Incompatibilities
Forms an explosive mixture with air.Contact with air causes the emission of toxic and corrosivefumes of selenium dioxide. A strong reducing agent. Reactsviolently with oxidizers, causing fire and explosion hazard.Incompatible with strong acids, water, halogenated hydro-carbons. Decomposes above100℃,forming toxic andflammable products including selenium and hydrogen.
Properties of HYDROGEN SELENIDE
| Melting point: | -65.73° |
| Boiling point: | bp -41.3° |
| Density | d4-42 2.12 |
| solubility | soluble in H2O |
| form | colorless gas |
| color | colorless gas; flammable |
| PH | 3.49(1 mM solution);2.93(10 mM solution);2.41(100 mM solution) |
| Water Solubility | mL/100mL H2O: 377 (4°C), 270 (22.5°C) [MER06]; mL/l00g H2O at standard temp, pressure: 386 (0°C), 351 (10°C), 289 (20°C) [LAN05] |
| Stability: | Stable. Highly flammable. Readily forms explosive mixtures with air. Incompatible with oxidizing agents, acids, water, halogenated hydrocarbons. |
| EPA Substance Registry System | Hydrogen selenide (7783-07-5) |
Safety information for HYDROGEN SELENIDE
Computed Descriptors for HYDROGEN SELENIDE
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid 1-Bromo-3,5-Di-Tert-Butylbenzene S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran
You may like
-
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1