GRAMICIDIN
- CAS NO.:1405-97-6
- Empirical Formula: C99H140N20O17
- Molecular Weight: 1882.2947
- MDL number: MFCD00131232
- EINECS: 215-790-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-15 16:23:17
What is GRAMICIDIN?
Description
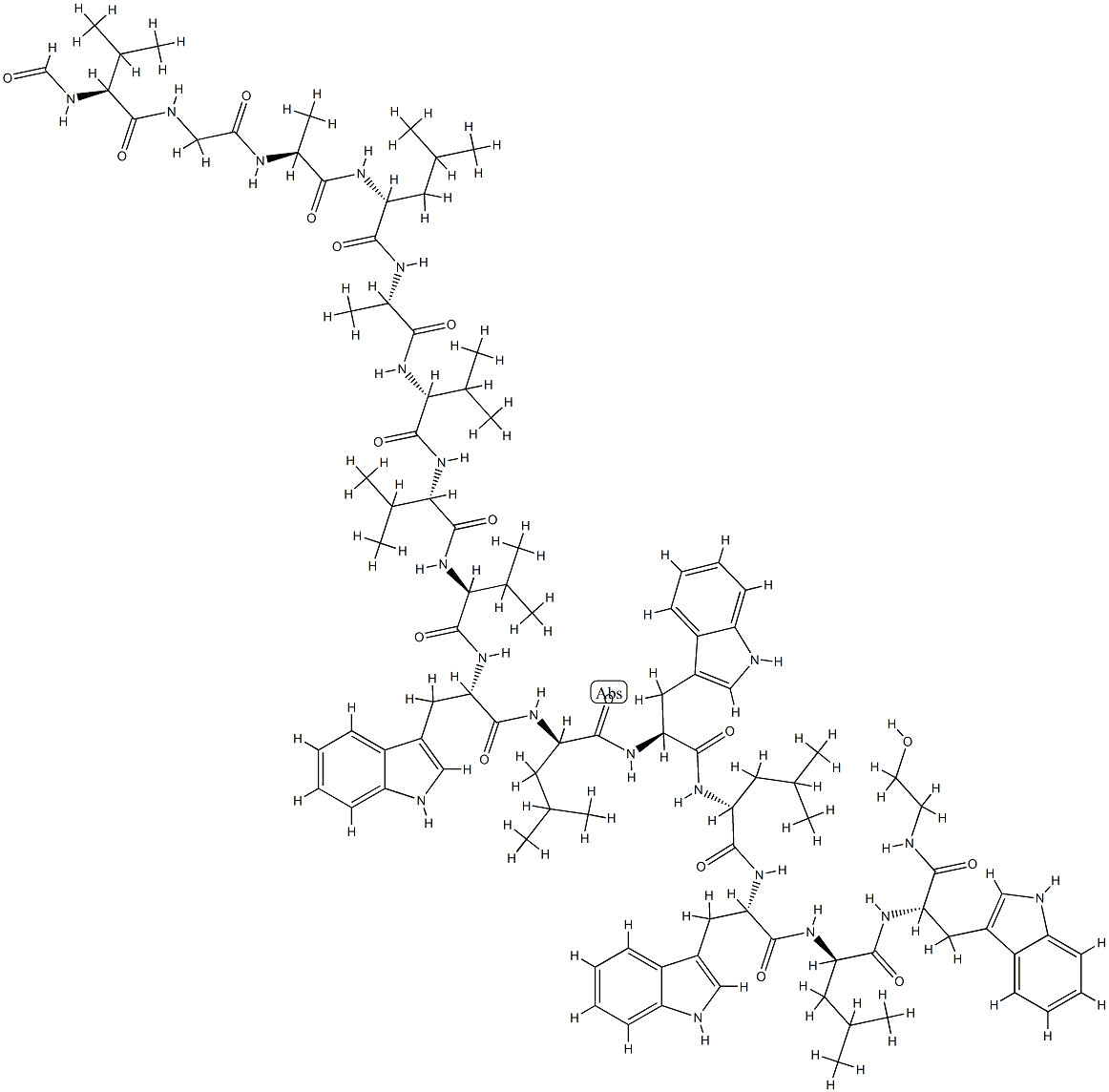
Gramicidin is a polypeptide antibiotic mixture of gramicidin A , gramicidin B, and gramicidin C originally isolated from B. brevis. It is also a component of the antibiotic tyrothricin . Gramicidin is active against the Gram-positive bacterium S. aureus but not Gram-negative E. coli. It also protects mice from Pneumococcus infection in vivo when administered at a dose of 0.002 mg/animal. Gramicidin inhibits growth of bacteria by forming pores and channels in the cell wall, which increases its permeability to monovalent cations. Formulations containing gramicidin have been used as topical agents in the treatment of bacterial skin infections.
Chemical properties
White, crystalline platelets. Soluble in lower alcohols, acetic acid, and pyridine; moderately soluble in dry acetone and dioxane; almost insoluble in water, ether, and hydrocarbons. Depresses surface tension, forms a fairly stable colloidal emulsion in distilled water.
The Uses of GRAMICIDIN
Gramicidins are a family of linear pentadecapeptides isolated from Bacillus brevis by Dubos at the Rockefeller Institute in 1939. Gramicidin is a complex of six closely related analogues A1, A2, B1, B2, C1 and C2 where A1 and A2 are the dominant components. Gramidicins act by forming channels in cell membranes causing ion leakage, and is an essential bioprobe for understanding the nature of the cell membranes.
The Uses of GRAMICIDIN
Medicine (antibacterial).
The Uses of GRAMICIDIN
Targets primarily the cell membrane of Gram-positive bacteria and shows little effect against Gram-negative bacteria. Gramicidin is typically used as a topical antimicrobial agent because it also causes hemolysis of red blood cells.
What are the applications of Application
Gramicidin from Bacillus aneurinolyticus (Bacillus brevis) is A linear polypeptide antibiotic
Background
Gramcidin D is a heterogeneous mixture of three antibiotic compounds, gramicidins A, B and C, making up 80%, 6%, and 14% respectively all of which are obtained from the soil bacterial species Bacillus brevis and called collectively gramicidin D. Gramcidins are 15 residue peptides with alternating D and L amino acids, which assemble inside of the hydrophobic interior of the cellular lipid bilayer to form a β-helix. Active against most Gram-positive bacteria and some Gram-negative organisms, Gramicidin D is used primarily as a topical antibiotic and is also found in Polysporin ophthalmic solution.
Indications
For treatment of skin lesions, surface wounds and eye infections.
Definition
An antibiotic produced by the metabolic processes of the bacteria Bacillus brevis. It is a polypeptide that is active against most Gram-positive pathogenic bacteria. It is one of the two antibiotic components of tyrothricin but has been isolated and used
brand name
Gramoderm (Schering).
Antimicrobial activity
Gramicidin is highly active against many Gram-positive bacteria. Neisseria spp. are relatively resistant. Gram-negative bacilli including Pseudomonas aeruginosa are susceptible although conflicting data exist about the degree of susceptibility. Gramicidin has a bactericidal activity against Mycoplasma spp. and several pathogenic fungi, including Candida albicans. Interestingly, it also appears to have antiviral activity against HIV and herpes simplex viruses (HSV-1, HSV-2).
General Description
Gramicidin is obtained from tyrothricin, a mixture ofpolypeptides usually obtained by extraction of cultures of B.brevis. Tyrothricin was isolated in 1939 by Dubos in aplanned search to find an organism growing in soil that wouldhave antibiotic activity against human pathogens. With onlylimited use in therapy now, it is of historical interest as thefirst in the series of modern antibiotics. Tyrothricin is a whiteto slightly gray or brown-white powder, with little or no odoror taste. It is practically insoluble in water and is soluble in alcoholand dilute acids. Suspensions for clinical use can beprepared by adding an alcoholic solution to calculatedamounts of distilled water or isotonic saline solutions.
Gramicidin acts as an ionophore in bacterial cell membranesto cause the loss of potassium ion from the cell. Itis bactericidal.Tyrothricin and gramicidin are effective primarilyagainst Gram-positive organisms. Their use is restricted tolocal applications. Tyrothricin can cause lysis of erythrocytes,which makes it unsuitable for the treatment of systemicinfections. Its applications should avoid direct contactwith the bloodstream through open wounds or abrasions. Itis ordinarily safe to use tyrothricin in troches for throat infections,as it is not absorbed from the GI tract. Gramicidinis available in various topical preparations containing otherantibiotics, such as bacitracin and neomycin.
Pharmaceutical Applications
Gramicidin as used in topical formulations is a mixture of
several closely related compounds, of which about 80% is in
the form of gramicidin A. It is part of the tyrothricin complex
originally isolated from B. brevis.
It is active against most species of Gram-positive bacteria,
including mycobacteria. Gram-negative bacilli are completely
insensitive.
It is highly toxic to erythrocytes, liver and kidney, and is
used only in topical formulations, usually as one of several
components.
Biochem/physiol Actions
Linear polypeptide antibiotic mixture of gramicidin A, B, C, and D. Gramicidin A acts as neutral carrier and helps in the establishment of ion flux across the lipid bilayer.
Mechanism of action
The mechanisms of gramicidin’s bactericidal activity have not been fully elucidated. Gramicidin is known to alter the function of the bacterial cytoplasmic membrane by forming channels that destroy the ion gradient and make it permeable for inorganic cations . It may also be a potent and specific inhibitor of the transcription reaction and inhibit the binding of DNA-dependent RNA polymerase (transcriptase) to DNA . Gramicidin appears to have antiviral activity against HIV, HSV-1, and HSV-2 viruses and is also used as a contraceptive due to its spermostatic properties.
Pharmacokinetics
Gramicidin is particularly effective against gram-positive bacteria. Because the drug is highly hemolytic, it cannot be administered internally and so is used only on the skin as a lotion or ointment. It is used primarily in the treatment of infected surface wounds, and in eye, nose, and throat infections. It is normally given with two other antibiotics (neomycin and polymixin B) as an ophthalmic solution.
Clinical Use
The use of gramicidin is restricted to topical applications on wounds or as ear and eye drops. Currently, much research is being invested to generate less toxic analogs that still exhibit the same wide range of bactericidal activity. To date, no new gramicidin-like polypeptides are available for clinical use. Gramicidin is used in some countries as a topical contraceptive, because it has spermostatic activity.
Metabolism
Not Available
Properties of GRAMICIDIN
| Melting point: | 229-230℃ |
| storage temp. | 2-8°C |
| solubility | Practically insoluble in water, soluble in methanol, sparingly soluble in alcohol |
| form | neat |
| form | Solid |
| color | White to Off-White |
| Water Solubility | Soluble in acetic acid and lower alcohols. Practically insoluble in water. |
| CAS DataBase Reference | 1405-97-6 |
Safety information for GRAMICIDIN
Computed Descriptors for GRAMICIDIN
| InChIKey | ZWCXYZRRTRDGQE-SORVKSEFSA-N |
GRAMICIDIN manufacturer
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran



![Gramicidin/CH63857,gramicidin from bacillus brevis,GRAMICIDIN D,GRAMICIDIN,GRAMICIDIN D [DUBOS],Gramicidin >95%](https://img.chemicalbook.in/)
![[L-α-Aminomyristic acid3,3']gramicidin S](https://img.chemicalbook.in/)
You may like
-
 1405-97-6 Gramicidine 99%View Details
1405-97-6 Gramicidine 99%View Details
1405-97-6 -
 Thermo Scientific Chemicals Gramicidin CAS 1405-97-6View Details
Thermo Scientific Chemicals Gramicidin CAS 1405-97-6View Details
1405-97-6 -
 Gramicidin CAS 1405-97-6View Details
Gramicidin CAS 1405-97-6View Details
1405-97-6 -
 3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details
3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details -
 20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 -
 3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details
3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details -
 57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 -
 4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
5305-40-8
