edoxaban
- CAS NO.:480449-70-5
- Empirical Formula: C24H30ClN7O4S
- Molecular Weight: 548.06
- MDL number: MFCD13195544
- EINECS: 859-181-7
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-20 21:15:22
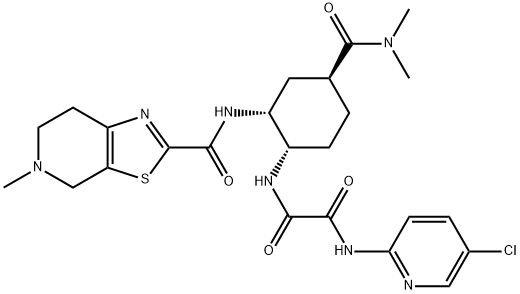
What is edoxaban?
Absorption
Following oral administration, peak plasma edoxaban concentrations are observed within 1-2 hours. Absolute bioavailability is 62%.
Toxicity
Premature discontinuation of any oral anticoagulant, including edoxaban, in the absence of adequate alternative anticoagulation increases the risk of ischemic events. If edoxaban is discontinued for reasons other than pathological bleeding or completion of a course of therapy, consider the use of another anticoagulant. Edoxaban increases the risk of potentially fatal major bleeding such as intracranial hemorrhage and gastrointestinal bleeding. Patients should be educated on how to watch for signs of major and minor bleeding and when to seek medical help. Co-administration of other anti-coagulants, anti-platelets, or thrombolytics may increase the risk of bleeding and should therefore be avoided.
The Uses of edoxaban
Edoxaban is an anticoagulant drug which acts as a direct factor Xa inhibitor.
The Uses of edoxaban
oral factor Xa inhibitor
Background
Edoxaban is a member of the Novel Oral Anti-Coagulants (NOACs) class of drugs, and is a rapidly acting, oral, selective factor Xa inhibitor. By inhibiting factor Xa, a key protein in the coagulation cascade, edoxaban prevents the stepwise amplification of protein factors needed to form blood clots. It is indicated to reduce the risk of stroke and systemic embolism (SE) in patients with nonvalvular atrial fibrillation (NVAF) and for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) following 5-10 days of initial therapy with a parenteral anticoagulant. Traditionally, warfarin, a vitamin K antagonist, was used for stroke prevention in these individuals but effective use of this drug is limited by it's delayed onset, narrow therapeutic window, need for regular monitoring and INR testing, and numerous drug-drug and drug-food interactions. This has prompted enthusiasm for newer agents such as dabigatran, apixaban, and rivaroxaban for effective clot prevention. In addition to once daily dosing, the benefits over warfarin also include significant reductions in hemorrhagic stroke and GI bleeding, and improved compliance, which is beneficial as many patients will be on lifelong therapy.
Indications
Edoxaban is indicated for reducing the risk of stroke and systemic embolism (SE) in patients with nonvalvular atrial fibrillation (NVAF). However, it should not be used in patients with creatinine clearance (CrCL) > 95 mL/min because of increased risk of ischemic stroke compared to warfarin at the highest dose studied (60 mg). It is also indicated for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) following 5-10 days of initial therapy with a parenteral anticoagulant.
Definition
ChEBI: A monocarboxylic acid amide that is used (as its tosylate monohydrate) for the treatment of deep vein thrombosis and pulmonary embolism.
Indications
Edoxaban is a direct oral anticoagulant (DOAC). It exerts its effects by inhibiting factor Xa (FXa). Edoxaban was FDA approved in early 2015 to treat deep venous thrombosis, pulmonary embolism and decrease the risk of hypercoagulability related illness; stroke, and systemic embolism (SE) in subjects with nonvalvular atrial fibrillation (NVAF).When compared to the popularly used anticoagulant warfarin, edoxaban entails more limited monitoring and possesses a reduced risk for significant bleeding and fewer interactions with other agents.Edoxaban is the most current direct oral anticoagulant (DOAC) and does not associate with the CYP-450 system.
Various clinical trials and studies (ENGAGE AF-TIMI and Hokusai-VTE) expressed edoxaban's effectiveness compared to the conventional vitamin K antagonist warfarin. It was demonstrated to be non-inferior in preventing blood clots when compared to warfarin. Edoxaban is the second FDA-approved anticoagulant agent with once-daily administration.Contrary to the other direct oral anticoagulants, apixaban and rivaroxaban, edoxaban has not yet received approval for secondary and postoperative prophylaxis for venous thromboembolism (VTE).
FDA-Approved Indications:
Deep venous thrombosis
Pulmonary embolism
Nonvalvular atrial fibrillation (NVAF)
https://www.ncbi.nlm.nih.gov/
Pharmacokinetics
Administration of edoxaban results in prolongation of clotting time tests such as aPTT (activated partial thromboplastin time), PT (prothrombin time), and INR (international normalized ratio).
Pharmacology
Similar to rivaroxaban and apixaban,edoxaban is an orally active, small-molecule (548 Da), reversible factor Xa inhibitor.As with the other direct oral factor Xa inhibitors,edoxaban exhibits a 10,000-fold greater selectivity for factor Xa compared with other serine proteins such as factor VlIa, t-PA, plasmin,or trypsin.Administered as edoxaban tosylate,the compound competitively inhibits free factor Xa directly without the need for antithrombin and factor Xa incorporated in the prothrombinase complex. The concentration-dependent inhibition of factor Xa leads to reduced thrombin generation and thrombin-induced platelet aggregation. Edoxaban inhibited factor Xa with Ki values of 0.561 nM for free factor Xa and 2.98 nM for prothrombinase.128 Edoxaban exhibits linear pharmacokinetics and produces concentration-dependent increases in the PT,INR, and aPTT.However,changes in these laboratory assays with edoxaban tend to be unpredictable and highly variable, reducing their use as a monitoring tool in clinical practice.
Pharmacokinetics and Pharmacodynamics of Edoxaban, a Non-Vitamin K Antagonist Oral Anticoagulant that Inhibits Clotting Factor Xa
Laboratory measurement of the anticoagulant activity of edoxaban: a systematic review
Pharmacokinetics, biotransformation, and mass balance of edoxaban, a selective, direct factor Xa inhibitor, in humans
Drug interactions
The majority of edoxaban pharmacokinetic drug interactions result from inhibition or induction of the P-gp efflux transporter,which is responsible for intestinal transport.Edoxaban taken with quinidine demonstrates an increase in edoxaban Cmax of 85% and AUS of 77%.Coadministration with dronedarone resulted in a Cmax and AUC increase of 46% and 85%,respectively.This drug interaction also increased the 24-h edoxaban concentration by 158%.Additionally, verapamil increased the edoxaban Cmax by 53%, the AUC by 53%, and the 24-h edoxaban concentration by 29%.146 As per the phase 3 clinical trial, patients receiving quinidine, dronedarone,or verapamil should receive the reduced dose of 30 mg daily instead of 60 mg daily.It should be noted that patients receiving azole antifungal agents, such as ketoconazole, or protease inhibitors were excluded from the phase 3 trial because of concerns about increased edoxaban exposure.Conversely, the use of rifampin, a P-gp inducer, resulted in a significant 34% reduction in the edoxaban AUC.Therefore, the combination of rifampin and edoxaban should be avoided.
Metabolism
Edoxaban is not extensively metabolized by CYP3A4 resulting in minimal drug-drug interactions. However, it does interact with drugs that inhibit p-gp (p-glycoprotein), which is used to transport edoxaban across the intestinal wall. Unchanged edoxaban is the predominant form in plasma. There is minimal metabolism via hydrolysis (mediated by carboxylesterase 1), conjugation, and oxidation by CYP3A4. The predominant metabolite M-4, formed by hydrolysis, is human-specific and active and reaches less than 10% of the exposure of the parent compound in healthy subjects. Exposure to the other metabolites is less than 5% of exposure to edoxaban.
Properties of edoxaban
| Melting point: | >213°C (dec.) |
| Density | 1.43 |
| storage temp. | Hygroscopic, Refrigerator, under inert atmosphere |
| solubility | Chloroform (Slightly), DMSO (Slightly, Heated), Methanol (Slightly, Heated) |
| form | Solid |
| pka | 9.46±0.70(Predicted) |
| color | White to Off-White |
Safety information for edoxaban
Computed Descriptors for edoxaban
| InChIKey | HGVDHZBSSITLCT-JLJPHGGASA-N |
| SMILES | C(NC1=NC=C(Cl)C=C1)(=O)C(N[C@H]1CC[C@H](C(N(C)C)=O)C[C@H]1NC(C1=NC2CCN(C)CC=2S1)=O)=O |
edoxaban manufacturer
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran
![(1R,2R,4S)-2-[(tert-butoxycarbonyl)aMino]-4-[(diMethylaMino)carbonyl]cyclohexyl Methanesulfonate](https://img.chemicalbook.in/CAS/20150408/GIF/929693-31-2.gif)
![Thiazolo[5,4-c]pyridine, 4,5,6,7-tetrahydro-5-methyl-](https://img.chemicalbook.in/CAS/GIF/259809-24-0.gif)
![(5H-Pyrrolo[3,4-d]thiazole-5-carboxylic acid,2-[[[(1R,2S,5S)-2-[[(5-chloro-1H-indol-2-yl)carbonyl]aMino]-5-[(diMethylaMino)carbonyl]cyclohexyl]aMino]carbonyl]-4,6-dihydro-,1,1-diMethylethyl ester](https://img.chemicalbook.in/CAS/20150408/GIF/365998-36-3.gif)
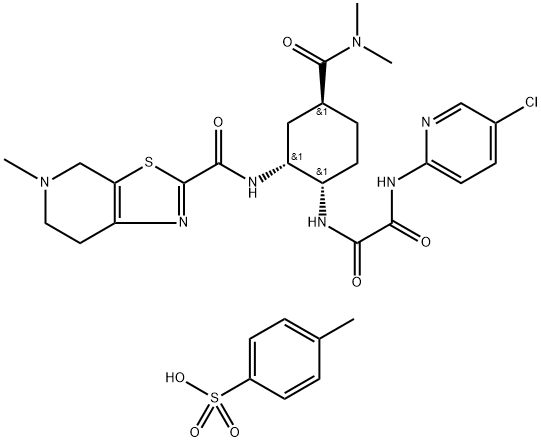
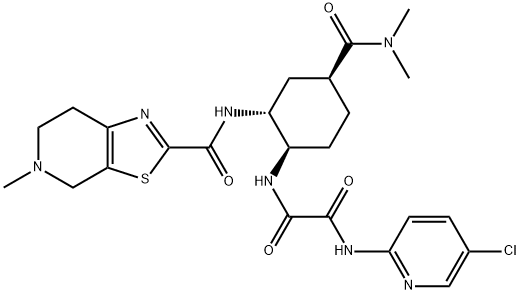
![2-BroMo-5-Methyl-4,5,6,7-tetrahydrothiazolo[5,4-c]pyridine](https://img.chemicalbook.in/CAS/GIF/143150-92-9.gif)
You may like
-
 480449-70-5 98%View Details
480449-70-5 98%View Details
480449-70-5 -
 Edoxaban 98%View Details
Edoxaban 98%View Details -
 Edoxaban 480449-70-5 98%View Details
Edoxaban 480449-70-5 98%View Details
480449-70-5 -
 480449-70-5 Edoxaban 99%View Details
480449-70-5 Edoxaban 99%View Details
480449-70-5 -
 480449-70-5 98%View Details
480449-70-5 98%View Details
480449-70-5 -
 Edoxaban 99%View Details
Edoxaban 99%View Details
480449-70-5 -
 Edoxaban 98%View Details
Edoxaban 98%View Details
480449-70-5 -
 Edoxaban 98% (HPLC) CAS 480449-70-5View Details
Edoxaban 98% (HPLC) CAS 480449-70-5View Details
480449-70-5
