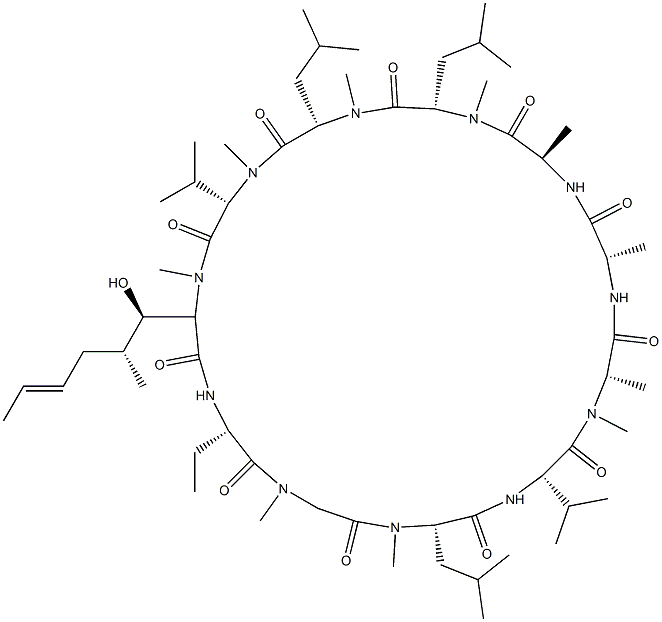
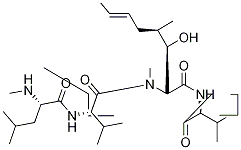
CYCLOSPORIN D
Synonym(s):1-(3-Chlorophenyl)piperazine-D8 hydrochloride solution
- CAS NO.:63775-96-2
- Empirical Formula: C63H113N11O12
- Molecular Weight: 1216.64
- MDL number: MFCD11111526
- EINECS: 806-434-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-06-30 15:45:59
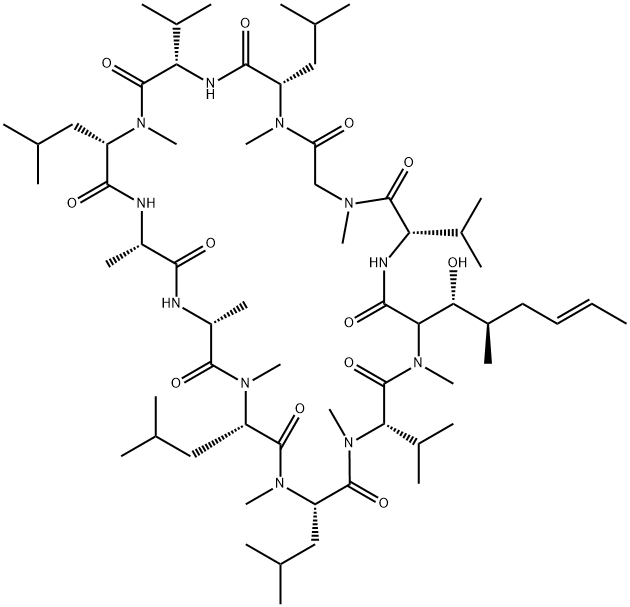
What is CYCLOSPORIN D?
Chemical properties
Colorless Crystalline Solid
The Uses of CYCLOSPORIN D
Cyclosporin D is a minor analogue of the cyclosporin family which is only weakly immunologically active. Cyclosporin D is a potent inhibitor of tumour-promoting phorbol esters on mouse skin in vivo, and a potent inhibitor of calcium/calmodulin-dependent EF-2 phosphorylation in vitro.
The Uses of CYCLOSPORIN D
A group of nonpolar cyclic oligopeptides with immunosupppressant activity.
What are the applications of Application
Cyclosporin D is a potent inhibitor of tumor promoting phorbol ester TPA/PMA
Biological Activity
cyclosporin d is an immunosuppressive agent [1].cyclosporin d (csd) is an analogue of cyclosporine a with weak immunosuppressive activity. cyclosporin d has been used as an internal standard for the quantification of cyclosporin a. in human multidrug-resistant ovarian cancer cells, cyclosporin d significantly overcame adriamycin resistance [2]. in lymphocyte, csd weakly inhibited pha-, pwm-, and pma + ca2+-induced cell proliferation [3].in mice, csd inhibited edema in mouse ear and alkaline phosphatase activity in mouse skin induced by tpa by 98% and 88%, respectively. in cytosol of mouse pancreas, csd inhibited the ca2+/calmodulin-dependent phosphorylation of the elongation factor 2 (ef-2) and the tpa-induced increase of ef-2 [1]. cyclosporin d was effective in inhibiting p. falciparum parasite in vitro and p. berghei malaria parasite development in vivo when administered orally [4].
storage
+4°C
References
[1]. gschwendt m, kittstein w, marks f. the weak immunosuppressant cyclosporine d as well as the immunologically inactive cyclosporine h are potent inhibitors in vivo of phorbol ester tpa-induced biological effects in mouse skin and of ca2+/calmodulin dependent ef-2 phosphorylation in vitro. biochem biophys res commun, 1988, 150(2): 545-551.
[2]. mizuno k, furuhashi y, misawa t, et al. modulation of multidrug resistance by immunosuppressive agents: cyclosporin analogues, fk506 and mizoribine. anticancer res, 1992, 12(1): 21-25.
[3]. sadeg n, pham-huy c, rucay p, et al. in vitro and in vivo comparative studies on immunosuppressive properties of cyclosporines a, c, d and metabolites m1, m17 and m21. immunopharmacol immunotoxicol, 1993, 15(2-3): 163-177.
[4]. uadia po1, ezeamuzie ic, ladan mj, et al. antimalarial activity of cyclosporins a, c and d. afr j med med sci, 1994, 23(1): 47-51.
Properties of CYCLOSPORIN D
| Melting point: | 148-1510C |
| Boiling point: | 1294.7±65.0 °C(Predicted) |
| alpha | D20 -245° (c = 0.52 in CHCl3); D20 -211° (c = 0.51 in methanol) |
| Density | 1.015±0.06 g/cm3(Predicted) |
| Flash point: | 9 °C |
| storage temp. | -20°C Freezer |
| solubility | Chloroform (Sparingly), Ethyl Aceatate (Slightly), Methanol (Slightly) |
| form | Colorless to off-white crystalline solid. |
| pka | 13.22±0.70(Predicted) |
| color | White to Off-White |
Safety information for CYCLOSPORIN D
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H225:Flammable liquids H319:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for CYCLOSPORIN D
| InChIKey | YJDYDFNKCBANTM-QCWCSKBGSA-N |
CYCLOSPORIN D manufacturer
Allmpus Laboratories Pvt Ltd
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid 1-Bromo-3,5-Di-Tert-Butylbenzene S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran
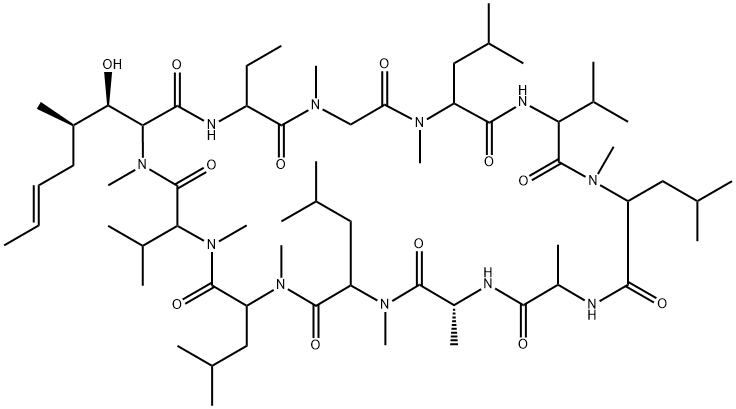
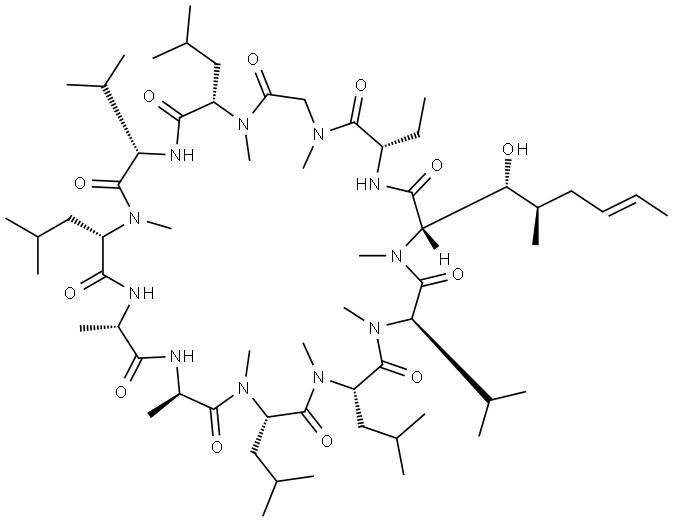
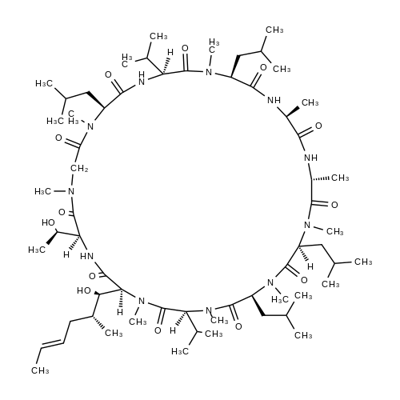

You may like
-
 63775-96-2 Cyclosporin D 96.06View Details
63775-96-2 Cyclosporin D 96.06View Details
63775-96-2 -
 Cyclosporin D 99.99View Details
Cyclosporin D 99.99View Details
63775-96-2 -
 Cyclosporin D solution CAS 63775-96-2View Details
Cyclosporin D solution CAS 63775-96-2View Details
63775-96-2 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1