Crotonaldehyde (trans predominantly)
- CAS NO.:123-73-9
- Empirical Formula: C4H6O
- Molecular Weight: 70.09
- MDL number: MFCD00007003
- EINECS: 204-647-1
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-17 09:49:40

What is Crotonaldehyde (trans predominantly)?
Chemical properties
Crotonaldehyde is water-white (turns paleyellow on contact with air) with an irritating, pungent, suffocating odor. The chemical can turn pale yellow when it contacts air. The chemical has a molecular weight of 70.1, a boiling point of 219°F, and a freezing point of -101°F. The vapor pressure is 30mm Hg at 25°C and the specific gravity is 0.87. The lower explosive limit is 2.1% and the upper explosive limit is 15.5%. Crotonaldehyde may be incompatible with caustics, ammonia, strong oxidizers, nitric acid, and amines. It also has the ability to polymerize at high temperatures.
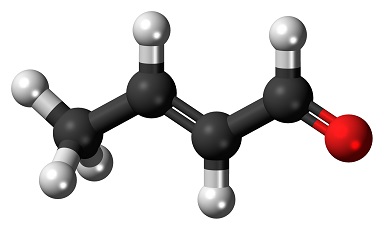
The Uses of Crotonaldehyde (trans predominantly)
Crotonaldehyde (2-butenal, β-methyl acrolein, propylene aldehyde) is similar in structure to acrolein, as both are α,β-unsaturated aldehydes. This structural similarity leads to similar sensitizing and irritating properties of the two compounds. Crotonaldehyde is used industrially in the preparation of other chemicals (chiefly sorbic acid), flavoring agents, and can form endogenously and in the environment.
Crotonaldehyde is used in the manufacture ofbutyl alcohol, butyraldehyde, and in severalorganic synthesis.
Definition
ChEBI: Crotonaldehyde is an enal consisting of propene having a formyl group at the 1-position.
General Description
Water-white to straw-colored liquid with a pungent, suffocating odor. Used as a chemical intermediate in a variety of industrial processes (surfactants, textiles, paper, fuels, insecticides, leather tanning, etc.). Used in chemical warfare.
Air & Water Reactions
Highly flammable.
Reactivity Profile
Crotonaldehyde is an aldehyde. Crotonaldehyde can react violently with strong oxidizing reagents, e.g., reaction with conc. nitric acid leads to instantaneous ignition [Andrussow, L., Chim. Ind. (Paris), 1961, 86, p. 542]. In contact with strong acids or bases Crotonaldehyde will undergo an exothermic condensation reaction. Reaction with 1,3-butadiene is particularly violent [Greenlee, K. W., Chem. Eng. News, 1948, 26, p. 1985]. Crotonaldehyde may rapidly polymerize with ethyl acetoacetate (Soriano, D.S. et al. 1988. Journal of Chemical Education 65:637.).
Health Hazard
Although slightly less toxic, crotonaldehyde is similar chemically and toxicologically to acrolein, which is rated as extremely toxic. Toxic concentrations for human inhalation have been reported at 12 mg/m3/10 minutes. Irritant dose to human eye is 45 ppm. As with acrolein, vapor exposures cause severe and painful eye irritation, damage to cornea, lacrimation (tearing), irritation of nasal membranes, pulmonary edema (filling of lungs with fluid) and gastrointestinal distress when ingested.
Health Hazard
Crotonaldehyde causes severe irritation ofthe eyes, nose, lungs, and throat. Exposureto a concentration of 12 mg/m3 in air for10 minutes can cause burning of the lungsand throat in humans. The symptoms ofinhalation toxicity in rats were excitement,behavioral change, convulsion, and death.The same symptoms were observed whencrotonaldehyde was administered subcutaneously.
LC50 value, inhalation (rats): 4000 mg/m3/30 minutes
LD50 value, subcutaneous (rats): 140 mg/kg
Crotonaldehyde is less toxic than acroleinor formaldehyde. The toxic symptoms, however,were similar to those of acrolein. Thecis-isomer of crotonaldehyde is mutagenic; itcaused cancer in test animals. Oral administrationof 2660 mg/kg for 2 years producedtumor in the liver in rats. Evidence of carcinogenicityin humans is not yet confirmed.
Fire Hazard
Vapors form explosive mixtures in air or in sewers. Hazardous peroxides and acids emitted when heated to decomposition. Avoid nitric acid. Unstable, avoid oxygen, heat, elevated pressures. Hazardous polymerization may occur. Avoid contact with alkaline materials such as caustic ammonia or amines, or at elevated temperatures.
Safety Profile
Suspected carcinogen. A poison by ingestion, subcutaneous, and intraperitoneal routes. Mutation data reported. A lachrymating material that is very dangerous to the eyes. Human respiratory system irritant by inhalation. Can cause corneal burns and is irritating to the skin. In case of contact, immediately flush the skin or eyes with water for at least 15 minutes and get medlcal attention. See also ALDEHYDES. Dangerous fire hazard when exposed to heat or flame. To fight fire, use alcohol foam, Con, dry chemical. Incompatible with 1,3-butadiene and oxidizing materials. When heated to decomposition it emits acrid smoke and fumes.
Potential Exposure
Crotonaldehyde is used as a warning agent in fuel gases and gas line leaks; as solvent; in Crotonaldehyde 935 chemical warfare; as an intermediate in the manufacture of n-butanol and crotonic and sorbic acids; in resin and rubber antioxidant manufacture; also used as a solvent in mineral oil purification; as an alcohol denaturant.
Shipping
UN1143 Crotonaldehyde or Crotonaldehyde, stabilized, Hazard class: 6.1; Labels: 6.1-Poison Inhalation Hazard, 3-Flammable liquid, Inhalation Hazard Zone B.
Incompatibilities
Vapors may form explosive mixture with air. A strong reducing agent. Readily converted by oxygen to peroxides and acids; heat or contact with alkalis and many other substances may cause polymerization. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides, nonoxidizing mineral acids; ammonia, organic amines; aliphatic amines; aromatic amines; 1,3-butadiene, strong bases. Liquid attacks some plastics, rubber, and coatings
Waste Disposal
Dissolve or mix the material with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. All federal, state, and local environmental regulations must be observed. May be absorbed on vermiculite and burned in open incinerator or dissolved in solvent and sprayed into incinerator
Properties of Crotonaldehyde (trans predominantly)
| Melting point: | −76 °C(lit.) |
| Boiling point: | 104 °C(lit.) |
| Density | 0.853 g/mL at 20 °C(lit.) |
| vapor density | 2.41 (vs air) |
| vapor pressure | 32 mm Hg ( 20 °C) |
| refractive index | n |
| Flash point: | 48 °F |
| storage temp. | 2-8°C |
| solubility | water: soluble425.4g/L at 20°C |
| form | Liquid |
| color | Clear |
| Odor | Pungent |
| explosive limit | 19.5% |
| Water Solubility | 150 g/L (20 ºC) |
| Merck | 14,2596 |
| BRN | 906731 |
| Exposure limits | TLV-TWA 6 mg/m3 (2 ppm)(ACGIH); IDLH
400 ppm (NIOSH). |
| Stability: | Light Sensitive |
| CAS DataBase Reference | 123-73-9(CAS DataBase Reference) |
| NIST Chemistry Reference | Crotonaldehyde(123-73-9) |
| EPA Substance Registry System | trans-Crotonaldehyde (123-73-9) |
Safety information for Crotonaldehyde (trans predominantly)
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Corrosion Corrosives GHS05  Skull and Crossbones Acute Toxicity GHS06  Health Hazard GHS08  Environment GHS09 |
| GHS Hazard Statements |
H225:Flammable liquids H315:Skin corrosion/irritation H318:Serious eye damage/eye irritation H330:Acute toxicity,inhalation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation H341:Germ cell mutagenicity H373:Specific target organ toxicity, repeated exposure H400:Hazardous to the aquatic environment, acute hazard |
| Precautionary Statement Codes |
P201:Obtain special instructions before use. P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P280:Wear protective gloves/protective clothing/eye protection/face protection. |
Computed Descriptors for Crotonaldehyde (trans predominantly)
| InChIKey | MLUCVPSAIODCQM-NSCUHMNNSA-N |
Crotonaldehyde (trans predominantly) manufacturer
JSK Chemicals
A.B. Enterprises
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran







You may like
-
 123-73-9 Crotonaldehyde, 98% 98%View Details
123-73-9 Crotonaldehyde, 98% 98%View Details
123-73-9 -
 Crotonaldehyde 98%View Details
Crotonaldehyde 98%View Details -
 crotonaldehyde 123-73-9 85%View Details
crotonaldehyde 123-73-9 85%View Details
123-73-9 -
 Crotonaldehyde, predominantly trans 99%View Details
Crotonaldehyde, predominantly trans 99%View Details
123-73-9 -
 Crotonaldehyde CAS 123-73-9View Details
Crotonaldehyde CAS 123-73-9View Details
123-73-9 -
 CROTONALDEHYDE For Synthesis CAS 123-73-9View Details
CROTONALDEHYDE For Synthesis CAS 123-73-9View Details
123-73-9 -
 Crotonaldehyde CASView Details
Crotonaldehyde CASView Details -
 CROTONALDEHYDEView Details
CROTONALDEHYDEView Details
123-73-9
