Celecoxib
Synonym(s):4-[5-(4-Methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzenesulfonamide;Celecoxib;YM-17;
- CAS NO.:169590-42-5
- Empirical Formula: C17H14F3N3O2S
- Molecular Weight: 381.37
- MDL number: MFCD00941298
- EINECS: 685-962-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-22 19:45:46
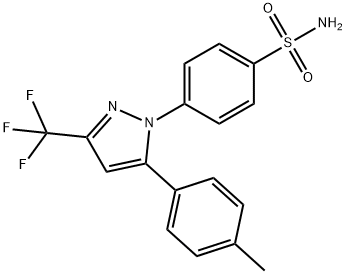
What is Celecoxib?
Absorption
Celecoxib is absorbed rapidly in the gastrointestinal tract. When a single oral dose of 200 mg was given to healthy research subjects, the peak plasma levels of celecoxib occurred within 3 hours. The Cmax is 705 ng/mL. When multiple doses are given, steady-state concentrations are reached on or before day 5. When taken with a high-fat meal, peak plasma levels are delayed for about 1 to 2 hours with an increase in total absorption (AUC) of 10% to 20%. The AUC of celecoxib has been shown to be significantly lower in patients with chronic renal impairment. A meta-analysis of pharmacokinetic studies has suggested an approximately 40% higher AUC (area under the curve) of celecoxib in black patients compared to Caucasians for unknown reasons.
Toxicity
The oral TDLo in humans 5.71 mg/kg.
It is not advisable to administer celecoxib in patients with renal impairment or advanced hepatic impairment, as this may lead to increased serum concentrations, causing toxicity. Symptoms of overdose may include breathing difficulties, coma, drowsiness, gastrointestinal bleeding, high blood pressure, kidney failure, nausea, sluggishness, stomach pain, and vomiting. Because serious gastrointestinal tract ulceration and bleeding can occur without preceding symptoms, patients should be monitored for signs/symptoms of gastrointestinal bleeding. Symptomatic and supportive measures should be taken in a celecoxib overdose. The induction of emesis or administration of active charcoal should take place if the patient is seen within 4 hours of celecoxib ingestion. Diuresis, urinary alkalinization, hemodialysis, or hemoperfusion may not be useful in a celecoxib overdose due to its high level of protein binding.
Description
Celecoxib is a nonsteroidal antiinflammatory drug (NSAID) first launched as Celebrex in the US for the treatment of symptoms in patients with rheumatoid arthritis (RA) and osteoarthritis (OA). Celecoxib belongs to a new class of 1, 5-diarylpyrazoles and can be synthesized by heat-promoted heterocyclization of a trifiuoro-l,3-dione with appropriate arylhydrazine. Celecoxib is a highly selective inhibitor of COX-2, the inducible form of cyclooxygenase expressed during inflammatory processes; it does not block the constitutive form COX-1, thus suppressing the gastric and intestinal toxicity of most non-selective NSAIDs. The potency ratio COX1/COX2 on purified human enzymes was about 400. In several in vivo models of acute and chronic inflammation, Celecoxib demonstrated potent antiinflammatory activity without affecting gastric or urinary prostaglandin PGE2. In several clinical studies performed with patients suffering from osteoarthritis or rheumatoid arthritis, Celecoxib was shown to be well tolerated and to relieve pain and inflammation more efficiently compared with other standard NSAIDs; the gastrointestinal safety profile was significantly better than that of other NSAIDs. Interestingly, Celecoxib was approved for another indication in patients with familial adenomatous polyposis (FAP). A six-month clinical trial demonstrated a 28% reduction in the number of colorectal polyps with Celecoxib, compared to a 5% reduction with placebo.
Chemical properties
White to Pale Yellow Solid
Originator
Searle (US)
The Uses of Celecoxib
A selective cyclooxygenase-2 (COX-2) inhibitor. Anti-inflammatory. Used in treatment of familial adenomatous polyposis
The Uses of Celecoxib
expectorant, gastric stimulant, insecticide
The Uses of Celecoxib
For relief and management of osteoarthritis (OA), rheumatoid arthritis (RA), juvenile rheumatoid arthritis (JRA), ankylosing spondylitis, acute pain, primary dysmenorrhea and oral adjunct to usual care for patients with familial adenomatous polyposis
Indications
Celecoxib is indicated for symptomatic treatment of adult osteoarthritis (OA) and adult rheumatoid arthritis (RA). Celecoxib is not a substitute for aspirin for cardiovascular event prophylaxis.
It may be also be used to treat acute pain from various sources, juvenile rheumatoid arthritis in children over 2, ankylosing spondylitis, and primary dysmenorrhea.
Celecoxib, in combination with tramadol, is indicated for the management of acute pain in adults severe enough to require an opioid analgesic and in whom alternative treatments are inadequate.
Background
Celecoxib, a selective cyclooxygenase-2 (COX-2) inhibitor, stands out among nonsteroidal anti-inflammatory drugs (NSAIDs) for its notably decreased risk of gastrointestinal bleeding compared to others in its class. Marketed by Pfizer under the brand name Celebrex, it gained FDA approval in 1998. This medication is primarily utilized to manage arthritis pain of various types and is also prescribed in familial adenomatous polyposis (FAP) to reduce precancerous polyps in the colon. Beyond its conventional applications, selective COX-2 inhibitors, particularly celecoxib, have attracted attention for their potential in cancer prevention and treatment. Clinical trials have explored their efficacy in combating various malignancies, highlighting their promising role in oncology.
Indications
Celecoxib is indicated for the treatment of osteoarthritis and rheumatoid arthritis. Its use is contraindicated in individuals with hypersensitivity to sulfonamides or other NSAIDs. It should be used with caution in persons with hepatic disease. Interactions occur with other drugs that induce CYP2C9 (e.g. rifampin rifampin) or compete for metabolism by this enzyme (e.g. fluconazole, leflunomide). The most common adverse reactions to celecoxib are mild to moderate GI effects such as dyspepsia, diarrhea, and abdominal pain. Serious GI and renal effects have occurred rarely.
Definition
ChEBI: Celecoxib is a member of the class of pyrazoles that is 1H-pyrazole which is substituted at positions 1, 3 and 5 by 4-sulfamoylphenyl, trifluoromethyl and p-tolyl groups, respectively. A cyclooxygenase-2 inhibitor, it is used in the treatment of arthritis. It has a role as a cyclooxygenase 2 inhibitor, a geroprotector, a non-steroidal anti-inflammatory drug and a non-narcotic analgesic. It is a member of toluenes, a sulfonamide, a member of pyrazoles and an organofluorine compound.
Manufacturing Process
4-(5-(4-Methylphenyl)-3-trifluoromethyl-N-pyrazol-1-yl)benzenesulfonamide To a solution of ethyl trifluoroacetate (1.90 ml, 16.0 mmol) in 7 ml of methyl tert-butyl ether was added 25% NaOMe (3.62 ml, 16.8 mmol). Next 4- chloroaceteophenone (2.08 ml, 16.0 mmol) in 2 ml of methyl tert-butyl ether was added. The mixture was stirred at room temperature overnight. To above solution was added 100 ml of 90% EtOH, followed by 4 N HCl (4.0 ml, 16 mmol) and 4-sulphonamidophenylhydrazine hydrochloride (3.58 g, 16 mmol). The mixture was heated to reflux for 3 hours. The mixture was concentrated. When 30 ml of water was added, a solid formed. The solid was filtered and washed with 20 ml of 60% EtOH to give 4.50 g of white solid. The filtrate was evaporated and taken up in ethyl acetate (100 ml), washed with saturated NaHCO3, and brine, dried over MgSO4, and concentrated. Heptane was added at boiling point of the mixture. After cooling down to 0°C, 1.01 g more product was obtained. The combined yield of the 4-(5-(4-methylphenyl)-3- trifluoromethyl-N-pyrazol-1-yl)benzenesulfonamide (Celecoxib) was 86%.
brand name
Celebrex (Searle).
Therapeutic Function
Antiinflammatory
General Description
Celecoxib (Celebrex) was the first selective COX-2 inhibitordrug introduced into the market in 1998 for use in thetreatment of RA, OA, acute pain, and menstrual pain. Thereal benefit is that it has caused fewer GI complicationswhen compared with other conventional NSAIDs. It hasalso been approved for reducing the number of adenomatouscolorectal polyps in familial adenomatous polyposis (FAP).Celecoxib is well absorbed and undergoes rapid oxidativemetabolism via CYP2C9 to give its inactive metabolites. Thus, a potential drug interaction exists betweencelecoxib and warfarin because the active isomer ofwarfarin is primarily degraded by CYP2C9.
Biological Activity
Selective cyclooxygenase-2 (COX-2) inhibitor (IC 50 values are 15 and 0.04 μ M for COX-1 and COX-2 respectively). Anti-inflammatory with shorter plasma half-life in vivo than SC 58121 (5-(4-Fluorophenyl)-1-[4-(methylsulfonyl)phenyl]-3-(trifluoromethyl)-1H-pyrazole ). Displays chemopreventive activity in in vivo tumor models.
Biochem/physiol Actions
Celecoxib is a non-steroidal, anti-inflammatory drug (NSAID) and a cyclooxygenase-2 (COX-2) selective inhibitor. Celecoxib is at least 10-20 times more selective for COX-2 over COX-1.
Pharmacokinetics
Celecoxib inhibits cyclooxygenase 2 (COX-2) enzyme, reducing pain and inflammation. It is important to note that though the risk of bleeding with celecoxib is lower than with certain other NSAIDS, it exists nonetheless and caution must be observed when it is administered to those with a high risk of gastrointestinal bleeding.
A note on the risk of cardiovascular events
Significant concerns regarding the safety of COX-2 selective NSAIDs emerged in the early 2000s. Rofecoxib, another member of the COX-2 inhibitor drug class, also known as Vioxx, was withdrawn from the market due to prothrombotic cardiovascular risks. Following an FDA Advisory Committee meeting in 2005, in which data from large clinical outcome trials were evaluated, the FDA concluded that the risk for cardiovascular thrombotic events for both COX-2 selective NSAIDs and nonselective NSAIDs was evident. It was determined that the benefits of celecoxib treatment, however, outweighed the risks. Postmarketing cardiovascular outcomes trial (PRECISION) revealed that the lowest possible dose of celecoxib was similar in cardiovascular safety to moderate strength doses of both naproxen and ibuprofen. Patients who had previous cardiovascular events including acute MI, coronary revascularization, or coronary stent insertion were not evaluated in the trial. It is not advisable to administer NSAIDS to these groups of patients.
Pharmacokinetics
Celecoxib is well absorbed from the GI tract, with peak plasma concentrations generally being attained within 3 hours of administration. Peak plasma levels in geriatric patients may be increased, but dosage adjustments in elderly patients generally are not required unless the patient weighs less than 50 kg.
Clinical Use
Celecoxib is currently indicated for the relief of signs and symptoms of osteoarthritis and rheumatoid arthritis and to
reduce the number of adenomatous colorectal polyps in familial adenomatous polyposis as an adjunct to usual care.
Celecoxib is synthesized by condensing 4-methyl-acetophenone and ethyltrifluoroacetate with sodium methoxide and
the resulting butanedione derivative cyclized with 4-hydrazinophenylsulfonamide. It was the first NSAID to be
marketed as a selective COX-2 inhibitor.
Synthesis
Celecoxib is prepared by condensation of 4-methylacetophenone with ethyl trifluoroacetate to give 4,4,4-trifluoro-1-(4- methylphenyl)butane-1,3-dione, which is cyclized with 4-hydrazinophenylsulfonamide.
in vitro
celecoxib not only reduced the production of pge2 but also inhibited the downstream effects of pge2. celecoxib blocked migration and invasion of a549 cells increased by pge2 in the wound healing and transwell assays. additionally, celecoxib reduced mmp9 mrna expression which was increased by pge2. moreover, celecoxib enhanced e-cadherin mrna expression which was inhibited by pge2 [2].
in vivo
celecoxib inhibited the increase in metastases of a549 cells and significantly reduced the increase in pge2 plasma level in mice receiving unilateral pneumonectomy [2].
Drug interactions
Potentially hazardous interactions with other drugs
ACE inhibitors and angiotensin-II antagonists:
antagonism of hypotensive effect; increased risk of
nephrotoxicity and hyperkalaemia.
Analgesics: avoid concomitant use of 2 or more
NSAIDs, including aspirin (increased side effects);
avoid with ketorolac (increased risk of side effects
and haemorrhage).
Antibacterials: possibly increased risk of convulsions
with quinolones; concentration reduced by
rifampicin
Anticoagulants: effects of coumarins and
phenindione enhanced; possibly increased risk of
bleeding with heparins, dabigatran and edoxaban -
avoid long term use with edoxaban.
Antidepressants: increased risk of bleeding with
SSRIs and venlaflaxine.
Antidiabetic agents: effects of sulphonylureas
enhanced
Antiepileptics: possibly increased phenytoin
concentration.
Antifungals: if used with fluconazole, halve the dose
of celecoxib.
Antivirals: increased risk of haematological toxicity
with zidovudine; concentration possibly increased by
ritonavir.
Ciclosporin: may potentiate nephrotoxicity
Cytotoxics: reduced excretion of methotrexate;
increased risk of bleeding with erlotinib.
Diuretics: increased risk of nephrotoxicity;
antagonism of diuretic effect; hyperkalaemia with
potassium-sparing diuretics.
Lithium: excretion decreased.
Pentoxifylline: possibly increased risk of bleeding
Tacrolimus: increased risk of nephrotoxicity.
The Uses of Celecoxib
Celecoxib is a nonsteroidal anti-inflammatory drug (NSAID) used to treat mild to moderate pain and help relieve symptoms of arthritis (eg, osteoarthritis, rheumatoid arthritis, or juvenile rheumatoid arthritis), such as inflammation, swelling, stiffness, and joint pain. Celecoxib is also used to treat ankylosing spondylitis, which is a type of arthritis that affects the joints in the spine. This medicine may also be used to treat acute pain and menstrual cramps. Celecoxib is also used to treat acute migraine headaches with or without aura. However, this medicine is not used to prevent migraines.
Metabolism
A large part of celecoxib metabolism is mediated by cytochrome P450 2C9 in the liver with some contribution from CYP3A4 and CYP2C8 and possible contributions from CYP2D6. It is metabolized by biotransformation to carboxylic acid and glucuronide metabolites. Three metabolites, a primary alcohol, a carboxylic acid, and a glucuronide conjugate, have been found in human plasma after celecoxib administration. These are considered inactive metabolites in regards to COX enzyme inhibition. Patients who are known or suspected to have decreased cytochrome P450 2C9 activity or function, based on their previous history, should be administered celecoxib with caution as they may have abnormally high serum concentrations resulting from decreased metabolism celecoxib.
Metabolism
Celecoxib is excreted in the urine and feces primarily as inactive metabolites, with less than 3% of an administered dose being excreted as unchanged drug. Metabolism occurs primarily in the liver by CYP2C9 and involves hydroxylation of the 4-methyl group to the primary alcohol, which is subsequently oxidized to its corresponding carboxylic acid, the major metabolite (73% of the administered dose). The carboxylic acid is conjugated, to a slight extent, with glucuronic acid to form the corresponding glucuronide. None of the isolated metabolites have been shown to exhibit pharmacological activity as inhibitors of either COX-1 or COX-2. Celecoxib also inhibits CYP2D6; thus, the potential of celecoxib to alter the pharmacokinetic profiles of other drugs inhibited by this isoenzyme exists. Celecoxib, however, does not appear to inhibit other CYP isoforms, such as CYP2C19 or CYP3A4. Other drug interactions related to the metabolic profile of celecoxib have been noted, particularly with other drugs that inhibit CYP2C.
Storage
Store at RT
References
1) Penning . (1997), Synthesis and biological evaluation of the 1,5-diarylpyrazole class of cyclooxygenase-2 inhibitors: identification of 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzenesulfonamide (SC-58635, celecoxib); J. Med. Chem., 40 1347 2) DeWitt et al. (1999), Cox-2-selective inhibitors: the new super aspirins; Mol. Pharmacol., 55 625 3) Harris et al. (2000), Chemoprevention of breast cancer in rats by celecoxib, a cyclooxygenase 2 inhibitor; Cancer Res., 60 2101 4) Koki and Masferrer et al. (2002), Celecoxib: a specific COX-2 inhibitor with anticancer properties; Cancer Control, 9 28
Properties of Celecoxib
| Melting point: | 157-159°C |
| Boiling point: | 529.0±60.0 °C(Predicted) |
| Density | 1.43±0.1 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | DMSO: >20mg/mL |
| form | powder |
| pka | 9.68±0.10(Predicted) |
| color | white to off-white |
| Water Solubility | 7mg/L(25 ºC) |
| Merck | 14,1956 |
| Stability: | Stable for 1 year from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20° for up to 3 months. |
| InChI | InChI=1S/C17H14F3N3O2S/c1-11-2-4-12(5-3-11)15-10-16(17(18,19)20)22-23(15)13-6-8-14(9-7-13)26(21,24)25/h2-10H,1H3,(H2,21,24,25) |
| CAS DataBase Reference | 169590-42-5(CAS DataBase Reference) |
| EPA Substance Registry System | Benzenesulfonamide, 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]- (169590-42-5) |
Safety information for Celecoxib
| Signal word | Danger |
| Pictogram(s) |
 Health Hazard GHS08  Environment GHS09 |
| GHS Hazard Statements |
H373:Specific target organ toxicity, repeated exposure H410:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P202:Do not handle until all safety precautions have been read and understood. P260:Do not breathe dust/fume/gas/mist/vapours/spray. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P391:Collect spillage. Hazardous to the aquatic environment P308+P313:IF exposed or concerned: Get medical advice/attention. |
Computed Descriptors for Celecoxib
| InChIKey | RZEKVGVHFLEQIL-UHFFFAOYSA-N |
| SMILES | C1(S(N)(=O)=O)=CC=C(N2C(C3=CC=C(C)C=C3)=CC(C(F)(F)F)=N2)C=C1 |
Celecoxib manufacturer
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran
![1-[4-[1-[4-(AMinosulfonyl)phenyl]-3-(trifluoroMethyl)-1H-pyrazol-5-yl]benzoate] β-D-Glucopyranuronic Αcid](https://img.chemicalbook.in/CAS/GIF/264236-79-5.gif)
You may like
-
 Celecoxib 99%View Details
Celecoxib 99%View Details -
 Celecoxib 99%View Details
Celecoxib 99%View Details -
 Celecoxib 98%View Details
Celecoxib 98%View Details -
 Celecoxib 98% CAS 169590-42-5View Details
Celecoxib 98% CAS 169590-42-5View Details
169590-42-5 -
 CAS NO 169590 42 5 CELECOXIB, 25Kg BagView Details
CAS NO 169590 42 5 CELECOXIB, 25Kg BagView Details
169590-42-5 -
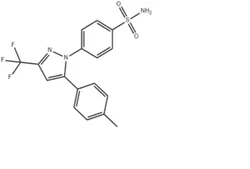 CELECOXIBView Details
CELECOXIBView Details
169590-42-5 -
 Celecoxib Api, 1 Kg BagView Details
Celecoxib Api, 1 Kg BagView Details
169590-42-5 -
 Celecoxib Powder APIView Details
Celecoxib Powder APIView Details
169590-42-5
