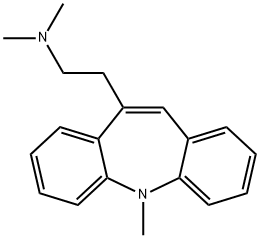
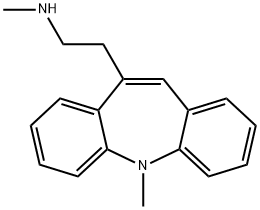
Amezepine
- CAS NO.:60575-32-8
- Empirical Formula: C18H20N2
- Molecular Weight: 264.36
- MDL number: MFCD00868469
- SAFETY DATA SHEET (SDS)
- Update Date: 2022-12-21 16:56:50
What is Amezepine?
Originator
Amezepine,ZYF Pharm Chemical
Manufacturing Process
78.0 g 9-methoxycarbony-10-methylacridinium methosulphate salt (obtained
according to a process analogous to that described by Rauhut et al. [J. Org.
Chem.,(30, 3587 (1965)], 800 ml ethanol and 40 ml distilled water are
mixed; then 80.0 g potassium borohydride are added over the course of 45
min, at room temperature. The mixture is agitated for 30 min, poure into a
water-ice mixture, agitated for 2 h, and extracted with ether; the extract is
dried by vacuum distillation. 51.0 g 9-methoxycarbonyl-10-methylacridan,
melting point 106°C (recrystallized from isopropyl ether) are obtained.
250.0 g 1-dimethylamino-2-chloroethane hydrochloride and 250 ml water are
mixed and cooled; 250 ml sodium hydroxide solution are added at a
temperature below 10°C; the mixture is agitated for 15 min at 5°C and
extracted with ether. The ethereal phases are washed with water and dried
over magnesium sulfate; the ether is distilled off and the residue is redistilled.
130.0 g 1-dimethylamino-2-chloroethane, boiling point 106°-108°C, are
obtained.
36.5 g 9-methoxycarbonyl-10-methylacridan and 1 L of toluene are mixed; 50
ml toluene are distilled off and the mixture is cooled. 5.56 g potassium are added at a temperature below +10°C; the mixture is agitated for 2 h at room
temperature, refluxed for 30 min, and cooled.
A solution of 52.5 ml 1-dimethylamino-2-chloroethane in 200 ml toluene is
added at a temperature below 20°C; the mixture is refluxed for 20 h and
cooled, and 100 ml t-butanol are added. The mixture is agitated for 1 h at
room temperature, 35 ml ethanol are added, and the mixture is again
agitated for 30 min at room temperature. It is then extracted with 2 N
hydrochloric acid; the acidic phases are washed with ether, made alkaline by
the addition of sodium hydroxide solution, and extracted with ether; the
ethereal phases are washed with water, dried over magnesium sulphate and
then distilled to dryness under a vacuum. The residue is dissolved in 100 ml
methanol; 300 ml of a saturated solution of fumaric acid in methanol are
added; the methanol is distilled off and replaced by ethyl acetate.
Crystallization is allowed to commence and the mixture is cooled with ice for
30 min and suction-filtered; the precipitate is washed with water and dried.
37.0 g 9-(2-dimethylaminoethyl)-9-methoxycarbonyl-10-methylacridan
fumarate are obtained, melting point 176°C (recrystallized from methanolethyl
acetate).
50.0 g of the 9-(2-dimethylaminoethyl)-9-methoxycarbonyl-10-methylacridan
fumarate are suspended in 500 ml water; the suspension is cooled and 30 ml
sodium hydroxide solution are added. The mixture is extracted with
dichloromethane and the organic phases are washed with water, dried over
magnesium sulphate and then distilled to dryness under a vacuum 36.0 g 9-
(2-dimethylaminoethyl)-9-methoxycarbonyl-10-methylacridan are obtained.
400 ml tetrahydrofuran are cooled and 20.0 g lithium aluminum hydride are
added over the course of 15 min, followed by a solution of 36.0 g 9-(2-
dimethylaminoethyl)-9-methoxycarbonyl-10-methylacridan in 400 ml
tetrahydrofuran added over the course of 30 min. The mixture is refluxed for
2 h and cooled, and 200 ml tetrahydrofuran containing 20% water are added
at a temperature below 0°C. The mixture is filtered, washed with
dichloromethane, and distilled to dryness under a vacuum; the residue is
dissolved in dichloromethane and the organic phases are washed with water,
dried over magnesium sulfate, and concentrated, ethyl acetate being added to
replace the dichloromethane. Crystallization is allowed to commence and the
mixture is cooled with ice for 1 h, suction-filtered, washed with water, and
dried.
24.5 g 9-(2-dimethylaminoethyl)-9-hydroxymethyl-10-methylacridan, melting
point 150°C (recrystallized from dichloromethane - ethyl acetate) are
obtained. On concentration of the mother liquors, a second yield of 1.6 g of
product is obtained (total yield: 79%).
A mixture of 24.5 g 9-(2-dimethylaminoethyl)-9-hydroxymethyl-10-
(methylacridan) 1 L of m-xylene and 125.0 g phosphorus pentoxide are
refluxed for 3 h; the reacture mixture is cooled, poured on to ice, agitated for
15 min, alkaline by the addition of sodium hydroxide solution, and extracted
with ethyl acetate; the organic phases are washed with water, dried over
magnesium sulfate, and dried by vacuum distillation. The residue is dissolved
in ether, filtered, and concentrated to a small volume; pentane is added with
distillation to remove the ether. The mixture is filtered and concentrated to
about 100 ml; crystallization is allowed to commence and the mixture is cooled with ice for 1 h and suction-filtered; the precipitate is washed with
water and dried in vacua. After chromatographing on magnesium silicate,
eluting with ether 16.0 g 5-methyl-10-(2-dimethylaminoethyl)-5Hdibenz[
b,f]azepine, melting point 78°C (recrystallized from pentane) are
obtained (yield: 70%).
32.5 ml benzene and 5 ml ethyl chloroformate are mixed; a solution of 6.5 g
5-methyl-10-(2-dimethylaminoethyl)-5H-dibenz[b,f] azepine in 32.5 ml
benzene are added; the mixture is refluxed for 5 h and cooled; ethyl acetate
is added and the mixture is agitated in the presence of N-hydrochloric acid;
the organic phase is decanted off, washed with water, dried over magnesium
sulfate, and distilled to dryness under a vacuum; 5.0 g 5-methyl-10-[2-(Nethoxycarbonyl-
N-methylamino)ethyl]-5H-dibenz[b,f]azepine are obtained.
(Yield: 64%).
5.0 g caustic potash in 50 ml n-butanol is added to 5.0 g 5-methyl-10-[2-(Nethoxycarbonyl-
N-methylamino)ethyl]-5H-dibenz[b,f]azepine; the mixture is
agitated and refluxed for 20 h, and the butanol is distilled off in vacua; the
residue is taken up with water, the mixture is extracted with ethyl acetate,
and the organic phases are washed with water and dried over magnesium
sulfate, and then distilled to dryness under vacuum; the residue is dissolved
in 10 ml ether and the solution is chromatographed on magnesium silicate,
eluting with ether, and distilled to dryness under a vacuum; 3.5 g crude 5-
methyl-10-(2-methylamino-ethyl)-5H-dibenz[b,f] azepine are obtained.
Therapeutic Function
Antidepressant
Properties of Amezepine
| Boiling point: | 411.6±34.0 °C(Predicted) |
| Density | 1.061±0.06 g/cm3(Predicted) |
| pka | 10.26±0.10(Predicted) |
Safety information for Amezepine
Computed Descriptors for Amezepine
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium 3-Bromopyrazole (3aR,4R,5R,6aS)-hexahydro-5-Triethyl silyloxy-4-((E)-3-oxo-5-phenylpent-1- enyl)cyclopenta[b]furan-2-one. 1-Chlorocarbonyl-4-piperidinopiperidine 1-Bromo-4-phenyl-2-Butanone 4-Amino-2-fluoro-N-methylbenzamide 1,1'-Carbonyldiimidazole SODIUM AAS SOLUTION ZINC AAS SOLUTION BUFFER SOLUTION PH 10.0(BORATE) GOOCH CRUCIBLE SINTERED AQUANIL 5 BERYLLIUM AAS SOLUTIONRelated products of tetrahydrofuran
You may like
-
![Dimethyl [2-oxo-3-[3-(trifluoromethyl)phenoxy]propyl]phosphonate 99%](https://img.chemicalbook.in//Content/image/CP5.jpg) Dimethyl [2-oxo-3-[3-(trifluoromethyl)phenoxy]propyl]phosphonate 99%View Details
Dimethyl [2-oxo-3-[3-(trifluoromethyl)phenoxy]propyl]phosphonate 99%View Details
54094-19-8 -
 85-81-4 99%View Details
85-81-4 99%View Details
85-81-4 -
 Cyclopentane carboxxylic acid 3400-45-1 99%View Details
Cyclopentane carboxxylic acid 3400-45-1 99%View Details
3400-45-1 -
![208111-98-2 (3aR,4R,5R,6aS)-5-(Benzoyloxy)hexahydro-4-[(1E)-3-oxo-4-[3-(trifluoromethyl)phenoxy]-1-buten- 1-yl]-2H-cyclopenta[b]furan-2-one 99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 208111-98-2 (3aR,4R,5R,6aS)-5-(Benzoyloxy)hexahydro-4-[(1E)-3-oxo-4-[3-(trifluoromethyl)phenoxy]-1-buten- 1-yl]-2H-cyclopenta[b]furan-2-one 99%View Details
208111-98-2 (3aR,4R,5R,6aS)-5-(Benzoyloxy)hexahydro-4-[(1E)-3-oxo-4-[3-(trifluoromethyl)phenoxy]-1-buten- 1-yl]-2H-cyclopenta[b]furan-2-one 99%View Details
208111-98-2 -
 2033-24-1 99%View Details
2033-24-1 99%View Details
2033-24-1 -
 Meldrums acid 2033-24-1 99%View Details
Meldrums acid 2033-24-1 99%View Details
2033-24-1 -
 Cyaclopentane carboxylic acid 99%View Details
Cyaclopentane carboxylic acid 99%View Details
3400-45-1 -
 2-Aminopyridine 504-29-0 99%View Details
2-Aminopyridine 504-29-0 99%View Details
504-29-0