368-16-1
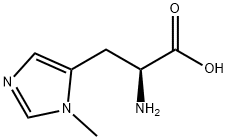
Product Name:
3-METHYL-L-HISTIDINE
Formula:
C7H11N3O2
Synonyms:
π-Methyl-L -histidine;1-Methyl-L -histidine (archaic);3-(1-Methylimidazol-5-yl)-L -alanine
Inquiry
CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Solid; [Sigma-Aldrich MSDS] |
|---|---|
| Solubility | 200 mg/mL at 25 °C |
| Collision Cross Section | 134.1 Ų [M+H]+ [CCS Type: DT, Method: single field calibrated with Agilent tune mix (Agilent)] |
| Chemical Classes | Other Uses -> Biochemical Research |
SAFETY INFORMATION
| Signal word | Warning |
|---|---|
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
COMPUTED DESCRIPTORS
| Molecular Weight | 169.18 g/mol |
|---|---|
| XLogP3 | -3.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Exact Mass | 169.085126602 g/mol |
| Monoisotopic Mass | 169.085126602 g/mol |
| Topological Polar Surface Area | 81.1 Ų |
| Heavy Atom Count | 12 |
| Formal Charge | 0 |
| Complexity | 174 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
N(pros)-methyl-L-histidine is a L-histidine derivative that is L-histidine substituted by a methyl group at position 3 on the imidazole ring. It has a role as a human metabolite and a Saccharomyces cerevisiae metabolite. It is a L-histidine derivative and a non-proteinogenic L-alpha-amino acid. It is a tautomer of a N(pros)-methyl-L-histidine zwitterion.