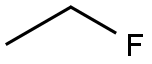
CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Ethyl fluoride appears as a colorless, odorless, flammable gas. Can cause asphyxiation by the displacement of air. Exposure of the container to prolonged heat or fire may cause it to rupture violently and rocket. |
|---|---|
| Chemical Classes | Solvents -> Chlorofluorocarbons |
SAFETY INFORMATION
| Signal word | Danger |
|---|---|
| Pictogram(s) |
 Flame Flammables GHS02  Gas Cylinder Compressed Gases GHS04 |
| GHS Hazard Statements |
H220:Flammable gases H280:Gases under pressure |
| Precautionary Statement Codes |
P260:Do not breathe dust/fume/gas/mist/vapours/spray. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. |
COMPUTED DESCRIPTORS
| Molecular Weight | 48.06 g/mol |
|---|---|
| XLogP3 | 0.9 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 0 |
| Exact Mass | 48.037528322 g/mol |
| Monoisotopic Mass | 48.037528322 g/mol |
| Topological Polar Surface Area | 0 Ų |
| Heavy Atom Count | 3 |
| Formal Charge | 0 |
| Complexity | 2.8 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Ethyl fluoride appears as a colorless, odorless, flammable gas. Can cause asphyxiation by the displacement of air. Exposure of the container to prolonged heat or fire may cause it to rupture violently and rocket.