219861-08-2
Product Name:
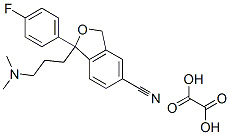
Escitalopram oxalate
Formula:
C22H23FN2O5
Synonyms:
Escitalopram oxalate;S-(+)-1-[3-(Dimethylamino)propyl]-1-(4-fluorophenyl)-1,3-dihydro-5-isobenzofurancarbonitrile oxalate;S-(+)-Citalopram oxalate
Inquiry
CHEMICAL AND PHYSICAL PROPERTIES
| Collision Cross Section | 178.8 Ų [M+H]+ [CCS Type: TW, Method: calibrated with polyalanine and drug standards] |
|---|
SAFETY INFORMATION
| Signal word | Warning |
|---|---|
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
COMPUTED DESCRIPTORS
| Molecular Weight | 414.4 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 6 |
| Exact Mass | 414.15910000 g/mol |
| Monoisotopic Mass | 414.15910000 g/mol |
| Topological Polar Surface Area | 111 Ų |
| Heavy Atom Count | 30 |
| Formal Charge | 0 |
| Complexity | 537 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 2 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Escitalopram Oxalate is the oxalate salt of escitalopram, a pure S-enantiomer of the racemic bicyclic phthalane derivative citalopram, with antidepressant activity. As a selective serotonin reuptake inhibitor (SSRI), escitalopram blocks the reuptake of serotonin by neurons in the central nervous system (CNS), thereby potentiating CNS serotonergic activity.
