SAFETY INFORMATION
| Signal word | Danger |
|---|---|
| Pictogram(s) |
 Skull and Crossbones Acute Toxicity GHS06 |
| GHS Hazard Statements |
H300:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P270:Do not eat, drink or smoke when using this product. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P332+P313:IF SKIN irritation occurs: Get medical advice/attention. P337+P313:IF eye irritation persists: Get medical advice/attention. P405:Store locked up. P403+P233:Store in a well-ventilated place. Keep container tightly closed. P501:Dispose of contents/container to..… |
COMPUTED DESCRIPTORS
| Molecular Weight | 306.4 g/mol |
|---|---|
| XLogP3 | 4.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 10 |
| Exact Mass | 306.18310931 g/mol |
| Monoisotopic Mass | 306.18310931 g/mol |
| Topological Polar Surface Area | 55.8 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 338 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
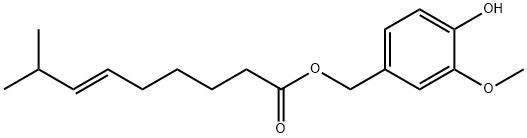
Capsiate is a carboxylic ester obtained by formal condensation of the carboxy group of (6E)-8-methylnon-6-enoic acid with the benzylic hydroxy group of vanillyl alcohol. A non-pungent analogue of capsaicin with a similar biological profile. It has a role as a plant metabolite, a hypoglycemic agent, an anti-allergic agent, an antioxidant, an angiogenesis inhibitor, an anti-inflammatory agent and a capsaicin receptor agonist. It is a carboxylic ester, a monomethoxybenzene and a member of phenols. It is functionally related to a vanillyl alcohol.