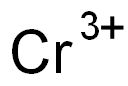CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Chromium nitrate appears as a purple-colored crystalline solid. Less dense than water. May ignite organic materials upon contact. Contact may irritate skin, eyes, and mucous membranes. Toxic by ingestion. Used to make other chemicals. |
|---|---|
| Chemical Classes | Metals -> Chromium Compounds, Inorganic |
SAFETY INFORMATION
| Signal word | Warning |
|---|---|
| Pictogram(s) |
 Flame Over Circle Oxidizers GHS03  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H272:Oxidising liquids;Oxidising solids H315:Skin corrosion/irritation H317:Sensitisation, Skin H319:Serious eye damage/eye irritation H411:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
COMPUTED DESCRIPTORS
| Molecular Weight | 238.01 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 0 |
| Exact Mass | 237.903958 g/mol |
| Monoisotopic Mass | 237.903958 g/mol |
| Topological Polar Surface Area | 189 Ų |
| Heavy Atom Count | 13 |
| Formal Charge | 0 |
| Complexity | 18.8 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 4 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Chromium nitrate appears as a purple-colored crystalline solid. Less dense than water. May ignite organic materials upon contact. Contact may irritate skin, eyes, and mucous membranes. Toxic by ingestion. Used to make other chemicals.