

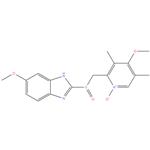
Esomeprazole EP Impurity E Omeprazole EP Impurity E ; Omeprazole N-Oxide ; Omeprazole USP Related Compound E 176219-04-8 96%
| Price | Get Latest Price | ||
| Packge | 25mg | 50mg | 100500100mg or 500 mg |
- Min. Order:100500100mg or 500 mg
- Time:2023-12-23
Product Details
- Product NameEsomeprazole EP Impurity E Omeprazole EP Impurity E ; Omeprazole N-Oxide ; Omeprazole USP Related Compound E
- CAS No.176219-04-8
- MFC17H19N3O4S
- MW361.42
- AppearanceSolidWhite to Off-White
- Melting point 173-175°C
- storage temp. Refrigerator
- density 1.40±0.1 g/cm3(Predicted)
- Boiling point 697.5±65.0 °C(Predicted)
Company Profile Introduction
KarpsChem is an independent research driven company specialized in the custom synthesis of pharmacopeia & non-pharmacopeia reference standards, drug gluconorides, drug metabolites, Isotope labelled reference standards and focused compound libraries. As a research-driven organization, we have developed novel processes to make niche products required to pharmaceutical companies globally at various platform. We understand the industry research needs thoroughly and have earned reputation for being most reliable innovator of pharmaceutical reference standards worldwide. Team having expertise of synthesis for undertaking CRAMs and CRO projects from Pharmaceutical, Agriculture and Veterinary industries. Variety of impurities like Genotoxic, Neurotoxic impurities required for agencies like USFDA, MHRDA etc,are available.
Our Expertise
All impurity standards will be supplied with certificates of analysis (COA) and NMR, Mass, HPLC purity. We offer custom synthesis of compounds on request. We provide technical support to your R&D.
Impurity Standards
We are supplier and manufacturer of impurity standards. Impurity standard certificates contain:
Identification by H-NMR and Mass spectroscopy. Purity test by either HPLC or GC. Other test like C NMR, TGA, C NMR, CHN, IR will be provide on request.
Custom synthesis mg to g
KARPSCHEM.SOLUTIONS offers world class service in chemical synthesis of a wide variety of organic compounds on the milligram to kilogram scale at competitive price.
Intermediates. Reference compounds. CRAM products.
Supplier other products
-

Albendazole EP Impurity A 5-(Propylthio)-1H-benzimidazol-2-amine
CAS:80983-36-4
Inquiry
-
Amlodipine EP impurity A Amlodipine USP RC D,Phthaloyl Amlodipine
CAS:88150-62-3
Inquiry
-
![Telmisartan Methyl Ester
2-(4-{[4-Methyl-6-(1-methyl-1H-1,3-benzodiazol-2-yl)
-2-propyl-1H-1,3-benzodiazol-1-yl]methyl}phenyl)benzoic acid
methyl ester](/ProductImageIndia/2024-03/Large/568eca52-0cb7-4799-b27b-c1a75464d9ce.png)
Telmisartan Methyl Ester 2-(4-{[4-Methyl-6-(1-methyl-1H-1,3-benzodiazol-2-yl) -2-propyl-1H-1,3-benzodiazol-1-yl]methyl}phenyl)benzoic acid methyl ester
CAS:528560-93-2
Inquiry
Recommended supplier
-
VIP0年
- Ebenezer Industries
- 176219-04-8 Omeprazole n-oxide 98%
- Inquiry
- 2024-03-30
-
VIP1年
- Orgamine Chemicals(I) Pvt Ltd
- Omeprazole n-oxide 98%
- Inquiry
- 2024-03-01
-
VIP1年
- Besil Chem LLP
- 176219-04-8 98%
- Inquiry
- 2024-01-29
-
VIP1年
- Varanous Labs Pvt Ltd
- 176219-04-8 omeprazole N-oxide 98%
- Inquiry
- 2023-12-26
- Since:2020-12-01
- Address: plot no-U107,Village nildoh,hingna midc,nagpur-440016.maharashtra,india
