

Safinamide
| Price | USD7.00 |
| Packge | 1KG |
- Min. Order:1KG
- Supply Ability:JD 669
- Time:2019-09-02
Product Details
- Product Name Safinamide
- CAS No. 133865-89-1
- EINECS No.603-772-2
- MFC17H19FN2O2
- MW302.34
- AppearancesolidWhite
- Melting point 208-212°
- Boiling point 476.7±40.0 °C(Predicted)
- density 1.189±0.06 g/cm3(Predicted)
- storage temp. under inert gas (nitrogen or Argon) at 2–8 °C
▼
▲
Product Name:
Synonyms:
SAFINAMIDE:(S)-(+)-2-[4-(3-FLUOROBENZYLOXY)BENZYLAMINO]PROPANAMIDE METHANSULFONATE;FCE 26743;(S)-(+)-2-[4-(3-Fluorobenzyloxy)benzylamino]propanamice methansulfonate;Safinamide Impurity D;133865-89-1;Safinamide;Safinamide ,Safinamide base;Safinamide Intermediate 2
CAS:
MF:
C17H19FN2O2
MW:
302.347
EINECS:
603-772-2
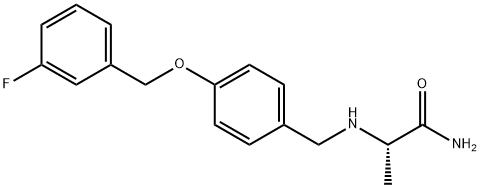
Mol File:
▼
▲
Safinamide Chemical Properties
▼
▲
Melting point
208-212°
Merck
14,8315
▼
▲
Safety Information
▼
▲
RTECS
TX1457385
▼
▲
Safinamide Usage And Synthesis
▼
▲
History
Safinamide is an α-aminoamide derivative with a multimodal mechanism of action involving both dopaminergic and non-dopaminergic properties. It increases levels of available dopamine through dopamine reuptake inhibition andMAO-B inhibition. It is a potent and reversible inhibitor of MAO-B, with significantly greater selectivity for MAO-B over MAO-A than selegiline and rasagiline. However, it also has an important and novel mode of action that involves blockade of Na+ channels andmodulation of Ca2+ channels that inhibits glutamate release and therefore may provide a level of cognitive improvement and neuroprotection. Homeostatic interactions between dopamine and glutamate are central to the normal physiology of the basal ganglia. In PD, this relationship is altered, resulting in upregulation of cortico-striatal glutamatergic function in l-dopa-induced dyskinesia. Any drug that can counteract such unbalance in glutamate function is potentially useful in controlling dyskinesias.
Safinamide was originated by Farmitalia Carlo Erba, a company that was later purchased by Pharmacia. Newron Pharmaceuticals was established as a spin-off of Pharmacia in 1999 and acquired safinamide rights and intellectual property from Pharmacia Corporation. Newron originally granted Serono exclusive worldwide rights to develop, manufacture, and commercialize safinamide in 2006. However, in October 2011, Merck Serono agreed to return full global rights for safinamide to Newron. Newron then finalized a strategic collaboration and license agreement with Zambon for the worldwide development and commercialization of safinamide.
Safinamide was originated by Farmitalia Carlo Erba, a company that was later purchased by Pharmacia. Newron Pharmaceuticals was established as a spin-off of Pharmacia in 1999 and acquired safinamide rights and intellectual property from Pharmacia Corporation. Newron originally granted Serono exclusive worldwide rights to develop, manufacture, and commercialize safinamide in 2006. However, in October 2011, Merck Serono agreed to return full global rights for safinamide to Newron. Newron then finalized a strategic collaboration and license agreement with Zambon for the worldwide development and commercialization of safinamide.
Indications
In 2003 positive preliminary results from a phase II trial of safinamide in epilepsy were announced. This open-labeled study was initiated to assess tolerability and drug–drug interaction (DDI) of safinamide in 48 patients with uncontrolled seizures that had already been treated with up to three other antiepileptic drugs. Starting with an initial oral dose of 50mg d−1, safinamide was increased every two weeks up to 300 mg d−1 or to the maximum tolerated dose. An interim analysis of the first 29 patients who completed the study showed excellent tolerability. In this group, no DDI was noted at any of the tested doses in that safinamide did not alter the kinetics of the other antiepileptic drugs. In this interim analysis safinamide was shown to be well tolerated in patients with medically intractable seizures. No serious AEs occurred in the study. Even though the study was not designed and powered to provide proof of efficacy, the sponsor reported that preliminary data showed a significant reduction in median seizure frequency from 50mg progressing up to the highest dose.
In 2005 safinamide was administered to a total of 10 patients with RLS that were enrolled into a single-center, phase II open-labeled pilot study. Each patient was administered safinamide (100 mg d−1) at bedtime for 2 weeks. A significant improvement in all efficacy parameters studied was observed when patients received safinamide. RLS is a neurological disorder characterized by jerky movements of the lower extremities that appear mostly in the evening and during sleep. As reported in a press release, safinamide was also found to be well tolerated and did not exhibit any clinically relevant side effects, but no study results have been published.
In 2005 safinamide was administered to a total of 10 patients with RLS that were enrolled into a single-center, phase II open-labeled pilot study. Each patient was administered safinamide (100 mg d−1) at bedtime for 2 weeks. A significant improvement in all efficacy parameters studied was observed when patients received safinamide. RLS is a neurological disorder characterized by jerky movements of the lower extremities that appear mostly in the evening and during sleep. As reported in a press release, safinamide was also found to be well tolerated and did not exhibit any clinically relevant side effects, but no study results have been published.
Mechanism of action
Safinamide may exert its in vivo effects through different mechanisms of action. It did not display activity against >80 different types of dopamine, glutamate, adenosine, serotonin, muscarinic, nicotinic, and GABA receptors. Conversely, potent modulation of DA metabolism, blockade of Na+/Ca2+ channels, and inhibition of glutamate release have been demonstrated. It has pointed out that the electrophysiological and neurochemical effects of safinamide are apparent at effective anticonvulsant concentrations. For example, pharmacokinetics (PK) showed that brain levels reach roughly 40 μM, at 30 and 60min after an oral dose of 10 mg kg?1 safinamide in rats. These points in time correspond to the peak anticonvulsant effect observed in the MES epilepsy model. This micromolar concentration approximates the concentration of safinamide that gives effective inhibition of excitatory amino acid release and reduction of sustained repetitive firing (SRF). Importantly, safinamide partitions itself well into the brain, where drug levels are approximately 10-fold higher than in plasma.
Company Profile Introduction
Henan CoreyChem Co., Ltd, based on the original Zhengzhou Cote Chemical Research Institute, be brave in absorbing highly educated talents & overseas returnees; actively responded to Zhengzhou City High-tech Zone Government’s Special Care Policy, reorganized and founded in National University of Science and Technology Park, which is a high-tech, stock enterprise of high-end chemical Custom synthesis;The park was created by the People's Government of Henan Province, and proved by Ministry of Education and the National Science & Technology, taking the construction mode of "many college a park, and common development", mainly depends on Zhengzhou University and Henan University’s scientific research and talent advantage to set up Universities, scientific research institute and enterprise scientific research achievements transformation platform, to make high-tech enterprises incubate, is the new high-tech talent gathering base, high and new technology industry enterprise radiation base, colleges and universities technological innovation base.
Henan Coreychem Co., Ltd, facing global High-tech pharmaceutical raw materials, high complex new type intermediates, fine chemicals custom synthesis, scale-up production and Rare chemicals trade. Corey have well-equipped machine, strong technical force and considerate marketing team service. We also have rich experience advantage in basic research, small scale process development, scale-up, industrial technology development & production and cost control.
Recommended supplier
-
VIP1年
- Shankus Pharmaceuticals
- 133865-89-1 Safinamide 98%
- Inquiry
- 2024-03-26
-
VIP1年
- Optimus Pharma Pvt Ltd (Sekhmet Pharmaventures))
- 133865-89-1 98%
- Inquiry
- 2024-02-28
- Since:2014-12-17
- Address: No.967,15th Floor,Unit 7, Building 1, No.70 of DianChang Road, High-tech Development Zone, Zhengzho
INQUIRY
