| Physical Description |
Solid |
| Color/Form |
Crystals from methanol-butanone |
| Melting Point |
142.5 °C |
| Solubility |
Slightly soluble |
| Stability/Shelf Life |
Commercially available fexofenadine hydrochloride capsules have an expiration date of 18 or 24 months after the date of manufacture when packaged in the manufacturer's unopened blister packages or high-density polyethylene bottles, respectively. Commercially available fexofenadine hydrochloride 30-mg conventional tablets have an expiration date of 18 months after the date of manufacture when packaged either in the manufacturer's unopened blister packages or high-density polyethylene bottles, whereas the commercially available fexofenadine hydrochloride 60-mg conventional tablets have an expiration date of 30 months after the date of manufacture when packaged either in the manufacturer's unopened blister packages or high-density polyethylene bottles. In addition, fexofenadine hydrochloride 180-mg conventional tablets have an expiration date of 18 or 30 months after the date of manufacture when packaged either in the manufacturer's unopened blister packages or high-density polyethylene bottles, respectively. |
| Collision Cross Section |
226.9 Ų [M+H]+ [CCS Type: TW, Method: Major Mix IMS/Tof Calibration Kit (Waters)] |
| Other Experimental Properties |
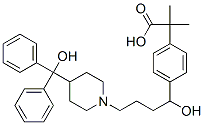
Molecular weight is 538.13. Fexofenadine hydrochloride is a white to off-white crystalline powder. It is freely soluble in methanol and ethanol, slightly soluble in chloroform and water, and insoluble in hexane. Fexofenadine hydrochloride is a racemate and exists as a zwitterion in aqueous media at physiological pH /Hydrochloride/ |