CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Solid |
|---|---|
| Melting Point | 220-224 °C |
| Solubility | 3.43 g/L |
| LogP | -0.2 |
| Collision Cross Section | 168.1 Ų [M+H]+ [CCS Type: TW, Method: calibrated with polyalanine and drug standards] |
SAFETY INFORMATION
| Signal word | Warning |
|---|---|
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H320:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
COMPUTED DESCRIPTORS
| Molecular Weight | 320.32 g/mol |
|---|---|
| XLogP3 | -0.2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 3 |
| Exact Mass | 320.12846858 g/mol |
| Monoisotopic Mass | 320.12846858 g/mol |
| Topological Polar Surface Area | 85.8 Ų |
| Heavy Atom Count | 23 |
| Formal Charge | 0 |
| Complexity | 521 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
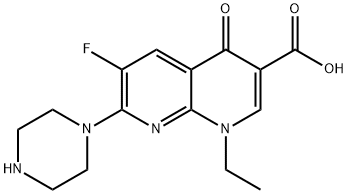
Enoxacin is a 1,8-naphthyridine derivative that is 1,4-dihydro-1,8-naphthyridine with an ethyl group at the 1 position, a carboxy group at the 3-position, an oxo sustituent at the 4-position, a fluoro substituent at the 5-position and a piperazin-1-yl group at the 7 position. An antibacterial, it is used in the treatment of urinary-tract infections and gonorrhoea. It has a role as an antibacterial drug and a DNA synthesis inhibitor. It is a monocarboxylic acid, an amino acid, a 1,8-naphthyridine derivative, a N-arylpiperazine, a quinolone antibiotic and a fluoroquinolone antibiotic.