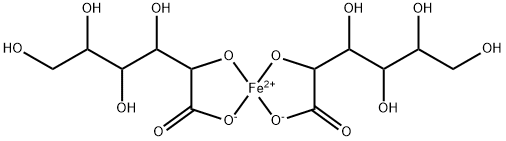
CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Dry Powder |
|---|---|
| Color/Form | The color of /ferrous gluconate/ solution depends on pH; they are light yellow at pH 2, brown at pH 4.5, and green at pH 7. The iron rapidly oxidizes at higher pH. |
| Melting Point | The melting point for D-gluconic acid, ferrous salt dihydrate (98%) is 188 °C, decomposes. The specific optical rotation is +6.7 deg at 25 °C (c=1, H2O). /Dihydrate/ |
| Solubility | Soluble with slight heating in water. Practically insoluble in ethanol |
| Stability/Shelf Life | Aqueous solutions are stabilized by the addition of glucose. |
| Decomposition | When heated to decomposition it emits acrid smoke and irritating fumes |
| pH | Between 4 and 5,5 (10 % solution) |
| Other Experimental Properties | Ascorbic acid and aminoacetic acid cause dark coloration; with pyridoxine, green color produced. |
COMPUTED DESCRIPTORS
| Molecular Weight | 466.17 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 13 |
| Hydrogen Bond Acceptor Count | 15 |
| Rotatable Bond Count | 10 |
| Exact Mass | 466.062106 g/mol |
| Monoisotopic Mass | 466.062106 g/mol |
| Topological Polar Surface Area | 278 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 385 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 8 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 4 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Ferrous Gluconate is a form of mineral iron for oral administration, Ferrous Gluconate is absorbed in the stomach and small intestine and combines with apoferritin to form ferritin, which is stored in the liver, spleen, red bone marrow, and intestinal mucosa. Important in transport of oxygen by hemoglobin to the tissues, iron is also found in myoglobin, transferrin, and ferritin, and is as a component of many enzymes such as catalase, peroxidase, and cytochromes. (NCI04)