112945-52-5
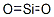
CHEMICAL AND PHYSICAL PROPERTIES
| Physical Description | Transparent to gray, odorless powder. Irritating to the skin and eyes on contact. Inhalation will cause irritation in the respiratory tract. [Note: Amorphous silica is the non-crystalline form of SiO2.] |
|---|---|
| Color/Form | Amorphous powder |
| Odor | Odorless |
| Boiling Point | 4046 °F at 760 mmHg (NIOSH, 2023) |
| Melting Point | 3110 °F (NIOSH, 2023) |
| Solubility | Insoluble (NIOSH, 2023) |
| Density | 2.2 (NIOSH, 2023) - Denser than water; will sink |
| Vapor Pressure | 0 mmHg (approx) (NIOSH, 2023) |
| Heat of Combustion | /Non-combustible/ |
| Other Experimental Properties | Amorphous silca: pyrogenic (fumed) amorphous silica (112945-52-5, formerly under 7631-86-9); Precipitated silica, including silica gel (112926-00-8, previously under 7631-86-9; deleted CAS number 112945-53-6); Diatomaceous earth (uncalcined)(61790-53-2; deleted CAS numbers 53571-43-0; 77108-41-9, 61970-41-0, 37337-67-0; 56748-40-4; 54990-62-4; 54990-61-3; 57692-84-9; 81988-94-5; 67417-47-4; 39455-02-2; 54511-18-1; 37264-95-2; 50814-24-9; 73158-38-0; 12623-98-2; 55839-10-6; 51109-72-9; 68368-75-2; 67016-73-3; 12750-99-1; 64060-29-3; 39421-62-0; 37328-66-8; 11139-66-5; 57126-63-3; 29847-98-1); Vitreous silica, quartz glass, fused silica (60676-86-0; deleted CAS numbers 55126-05-1; 119573-97-6; 37224-35-4; 37224-34-3); Flux-calcined diatomaceous earth (68855-54-9) |
| Chemical Classes | Mineral Dusts -> Other Mineral Dusts |
COMPUTED DESCRIPTORS
| Molecular Weight | 60.084 g/mol |
|---|---|
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 59.966755773 g/mol |
| Monoisotopic Mass | 59.966755773 g/mol |
| Topological Polar Surface Area | 34.1 Ų |
| Heavy Atom Count | 3 |
| Formal Charge | 0 |
| Complexity | 18.3 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
PRODUCT INTRODUCTION
description
Silica is another name for the chemical compound composed of silicon and oxygen with the chemical formula SiO2, or silicon dioxide. There are many forms of silica. All silica forms are identical in chemical composition, but have different atom arrangements. Silica compounds can be divided into two groups, crystalline (or c-silica) and amorphous silica (a-silica or non-crystalline silica). c-Silica compounds have structures with repeating patterns of silicon and oxygen. a-Silica chemical structures are more randomly linked when compared to c-silica. All forms of silica are odorless solids composed of silicon and oxygen atoms. Silica particles become suspended in air and form non-explosive dusts. Silica may combine with other metallic elements and oxides to form silicates.
